Discovering cell surface proteins that are specific to cancer cells and not normal cells can create opportunities for safer delivery of toxic drugs to tumors. Delaveris et al. identified a family of proteins that are delivered to the cell surface in cancer but not in healthy tissues (see the Perspective by Pfannenstein and Meyer). One such protein is the cancer-promoting enzyme Src. The researchers developed antibodies that target the surface form of Src and showed that these anti-Src antibodies could specifically deliver toxic radioactive isotopes or recruit killer T cells to tumors in vivo. This work highlights the potential to increase the types of cell surface targets available for cancer therapies. —Stella M. Hurtley
Antibody-targeted therapeutics for cancer (e.g., T cell engagers, antibody-based radioligand therapies, and antibody-drug conjugates) require a cancer-associated protein to be displayed on the cell surface at much higher levels than in healthy tissues. Typically, targets are proteins highly up-regulated by cancer cells for survival (e.g., HER2 and EGFR). However, therapies targeting these proteins are limited by the expression of these cell surface targets in healthy tissues. There is therefore a need to find and understand new classes of tumor-associated cell surface proteins that have greater differences in tumor versus healthy tissue expression. Interest in new targets for cancer therapeutics has led to the discovery of new ways to differentiate tumor cells from healthy cells using their cell surface proteins. These differences include genetic mutations specific to cancer, the activities of enzymes yielding new posttranslational modifications, and aberrant protein localization.
We developed a system to identify cell surface kinase substrates as well as to define a high-resolution cell surface proteome through photo-proximity labeling proteomics. This method allowed us to both identify protein substrates that get phosphorylated on the cell surface and gain insights into the enzymes mediating phosphorylation.
In this work, we found that Src, the master regulatory kinase typically anchored by its N-myristoyl lipid to the inner leaflet of the plasma membrane, was noncanonically translocated to the cell surface through autophagolysosomal exocytosis (ALE). We found additional evidence that other N-myristoylated intracellular proteins could be translocated in a similar fashion. Using cell lines with varying levels of cellular Src (cSrc), we observed that the transport of Src to the cell surface was regulated both by autolysosomal flux and total Src protein level. We observed that extracellular Src (eSrc) was present on the surface of cancer cell lines, whole and disaggregated xenografts, and disaggregated patient tumor samples. We propose that the prevalence of eSrc on cancer cells is likely attributable to both cSrc protein up-regulation in cancer and dysregulated autophagic flux, a hallmark of cancer, leading to translocation and inversion of cSrc by ALE. Additionally, we characterized the cell surface phosphotyrosine substrates of eSrc and showed that eSrc activity promoted cell proliferation. Finally, we demonstrated the translational potential of eSrc as a diagnostic and therapeutic cancer antigen using an anti-Src antibody formulated as a bispecific T cell engager (TCE), an antibody-drug conjugate (ADC), or a radioligand therapeutic (RLT).
We describe a class of mislocalized proteins, prototypically the proto-oncogene tyrosine kinase Src. These proteins are N-terminal lipid-modified proteins, canonically anchored to the plasma membrane, that undergo topological inversion through a noncanonical secretory pathway that we elucidate in this work. Collectively, these findings uncover a class of proteins by which cancer cells can be therapeutically targeted with antibodies.
Overexpression of the proto-oncogene Src is common to a wide variety of cancers. In this work, we found that Src is noncanonically translocated and inverted onto the cell surface in cancer, both in vitro and in vivo. We identified autophagolysosomal exocytosis (ALE) as a secretory mechanism prominent in cancer cell lines. Src represents the prototypical example of a family of membrane-anchored proteins that are transported by this process. Furthermore, this extracellular membrane–associated Src (eSrc) was found in primary tumors, and anti-Src antibody-based therapies mediated tumor cell killing in cell culture systems and in mouse xenograft models. Thus, intracellular N-myristoylated proteins, prototypically Src, can be topologically inverted onto the cell surface in cancer and targeted with antibody therapeutics.
发现癌细胞而非正常细胞特有的细胞表面蛋白可以为更安全地将毒性药物递送至肿瘤创造机会。Delaveris等人鉴定了一类在癌症中递送到细胞表面但在健康组织中不存在的蛋白质家族(参见Pfannenstein和Meyer的观点文章)。其中一种蛋白是促进癌症的酶Src。研究人员开发了针对Src表面形式的抗体,并表明这些抗Src抗体能够特异性递送毒性放射性同位素或在体内招募杀伤性T细胞到肿瘤。这项工作突显了增加可用于癌症疗法的细胞表面靶点类型的潜力。 —Stella M. Hurtley
针对癌症的抗体靶向疗法(例如T细胞衔接器、基于抗体的放射性配体疗法和抗体-药物偶联物)要求癌症相关蛋白在细胞表面的表达水平远高于健康组织。通常,靶点是癌细胞为生存而高度上调的蛋白(例如HER2和EGFR)。然而,针对这些蛋白的疗法受到这些细胞表面靶点在健康组织中表达的限制。因此,需要发现和理解新的肿瘤相关细胞表面蛋白类别,这些蛋白在肿瘤与健康组织表达中的差异更大。对癌症疗法新靶点的兴趣促使人们发现了利用细胞表面蛋白区分肿瘤细胞与健康细胞的新方法。这些差异包括癌症特异的基因突变、产生新翻译后修饰的酶活性以及异常的蛋白定位。
我们开发了一个系统,通过光邻近标记蛋白质组学来鉴定细胞表面激酶底物,并定义高分辨率的细胞表面蛋白质组。这种方法使我们能够鉴定在细胞表面被磷酸化的蛋白质底物,并深入了解介导磷酸化的酶。
在这项工作中,我们发现Src,一种通常通过其N-肉豆蔻酰脂锚定在质膜内小叶的主调控激酶,通过自噬溶酶体胞吐作用(ALE)非经典地易位到细胞表面。我们发现了额外的证据,表明其他N-肉豆蔻酰化的细胞内蛋白可能以类似的方式易位。使用具有不同细胞Src(cSrc)水平的细胞系,我们观察到Src向细胞表面的运输受自噬溶酶体通量和总Src蛋白水平的调节。我们观察到细胞外Src(eSrc)存在于癌细胞系、完整和分散的异种移植瘤以及分散的患者肿瘤样本表面。我们提出eSrc在癌细胞上的普遍存在可能归因于癌症中cSrc蛋白的上调和自噬通量失调(癌症的一个标志),导致cSrc通过ALE易位和翻转。此外,我们表征了eSrc的细胞表面磷酸酪氨酸底物,并表明eSrc活性促进了细胞增殖。最后,我们证明了eSrc作为诊断和治疗性癌症抗原的转化潜力,使用配制成双特异性T细胞衔接器(TCE)、抗体-药物偶联物(ADC)或放射性配体疗法(RLT)的抗Src抗体。
我们描述了一类错误定位的蛋白,典型代表是原癌基因酪氨酸激酶Src。这些蛋白是N端脂质修饰蛋白,通常锚定在质膜上,通过我们在这项工作中阐明的一种非经典分泌途径发生拓扑翻转。总的来说,这些发现揭示了一类蛋白,通过它们可以用抗体对癌细胞进行治疗性靶向。
原癌基因Src的过表达在多种癌症中很常见。在这项工作中,我们发现Src在癌症中非经典地易位并翻转到细胞表面,无论是在体外还是体内。我们确定了自噬溶酶体胞吐作用(ALE)是癌细胞系中突出的分泌机制。Src代表了通过这一过程运输的膜锚定蛋白家族的典型例子。此外,这种细胞外膜相关Src(eSrc)在原发肿瘤中被发现,基于抗Src抗体的疗法在细胞培养系统和小鼠异种移植模型中介导了肿瘤细胞杀伤。因此,细胞内N-肉豆蔻酰化蛋白(典型代表Src)在癌症中可以拓扑翻转到细胞表面,并可用抗体疗法进行靶向。
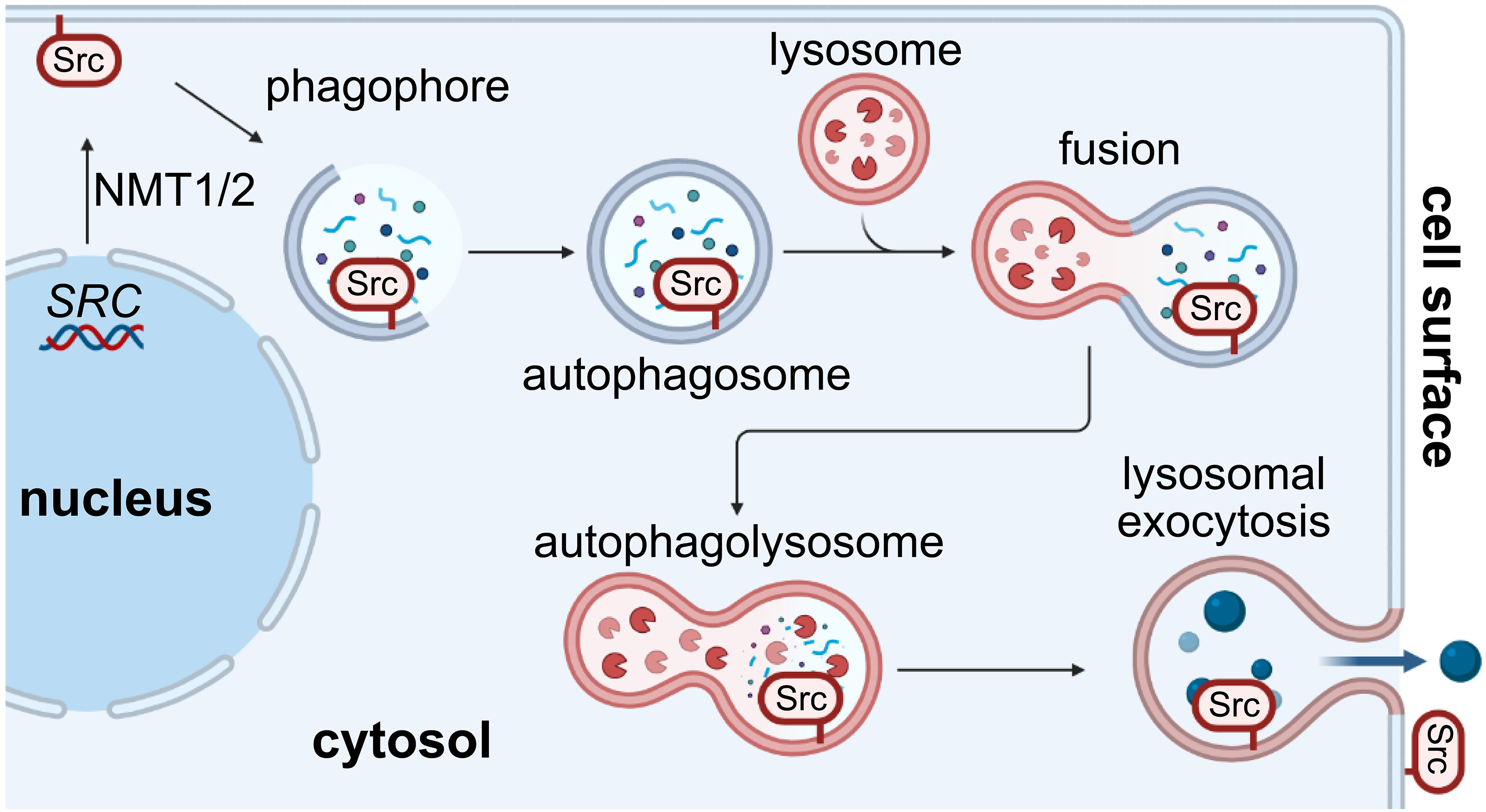
图:Src被转运至细胞表面。SRC经过翻译后,蛋白质发生脂质修饰,脂质部分插入质膜的内叶。成核的吞噬泡膜起源于质膜成分,同时携带Src。自噬体形成后,与溶酶体融合产生自噬溶酶体,这些自噬溶酶体可通过胞吐作用将Src呈现于细胞表面。[该图部分使用BioRender.com创建]
Autophagolysosomal exocytosis inverts Src kinase onto the cell surface in cancer