Tumor-resident stem cells have been suggested to facilitate cancer progression and long-term recurrence, but their identity and functions in gastric cancer remain unclear. Lim et al. report that the surface marker AQP5 enriches for gastric cancer stem cells in mouse and human tumors arising from the distal pyloric stomach. AQP5+ stem cells reestablished tumors after transplantation and generated long-lived organoid cultures. Selective ablation of AQP5 was associated with tumor regression in experimental model systems. AQP5 expression on stem cells modulated WNT, PI3K, and MAPK signaling and enhanced tumor proliferation in several experimental models of gastric cancer. —Priscilla N. Kelly
Cancer stem cells (CSCs) are self-renewing populations capable of generating the differentiated tumor bulk and sustaining the long-term maintenance of these cancers. These traits confer CSCs with the ability to drive sustained tumor progression as well as tumor recurrence after cancer treatment. The ability to define and selectively eliminate CSC populations holds potential as a therapeutic strategy. CSCs have been identified in a number of cancers from the blood, brain, and colon, but a robust investigation and validation of their identity in gastric tumors have remained elusive to date, precluding an examination of CSC roles in this context.
Despite its biological and therapeutic implications, several major obstacles stand in the way of identifying robust surface markers of CSCs in gastric cancers: (i) Many CSC markers display broad expression across cell and tissue types, limiting their utility in isolating these populations; (ii) most markers are poorly validated, relying on marker associations rather than functional assays that provide a readout of stem cell potential; and (iii) many models lack sufficient physiological relevance in recapitulating the molecular and morphological features present in human cancers. There is therefore a need to develop a unified strategy to identify CSC markers in gastric tumors that may hold potential for therapeutic use.
We developed methods to isolate aquaporin-5–positive (AQP5+) CSCs from mouse and human pyloric tumor tissues, including metastatic tumor samples. Transcriptomic profiling revealed that AQP5+ tumor cells were enriched in a subset of epithelial cells expressing genes associated with stem cell activity. We confirmed CSC potential using multiple functional assays demonstrating that mouse and human AQP5+ cells were able to initiate long-lived organoid cultures and reestablish invasive tumors when transplanted into mice, whereas AQP5− cells failed to do so. In line with these findings, targeted ablation of AQP5+ cells blocked tumor initiation and drove tumor regression in established tumors, which could be eliminated using a repeated ablation strategy to circumvent tumor plasticity. From a functional standpoint, we found that AQP5 expression in these gastric CSCs promoted cancer cell proliferation and invasion through WNT, PI3K (phosphatidylinositol 3-kinase), and MAPK (mitogen-activated protein kinase) signaling across diverse, near-physiological organoid and mouse models of gastric cancer, highlighting the broader relevance of our findings.
We present identification of a surface marker of gastric CSCs that also plays a functional role in driving cancer progression and metastasis. Unlike previously proposed markers, AQP5 robustly enriches for CSCs within a specific subset of gastric cancer cells in both mice and humans, where it also promotes key cancer traits. These findings highlight possibilities for examining CSC-niche contributions in a variety of contexts, including tumor plasticity, recurrence, and therapeutic drug resistance, and may assist the development of targeted therapies against AQP5+ CSCs in gastric cancers.
Cancer stem cells (CSCs) represent a self-renewing population capable of fueling long-term tumor growth. In gastric cancer, the identity of CSC populations remains unclear. In this study, we established a gastric CSC population marked by the water channel protein aquaporin-5 (AQP5), which resides in human and mouse pyloric tumors. Using multiple organoid and mouse models, we found a requirement for AQP5+ CSCs in both initiating and sustaining cancer progression and demonstrated that AQP5 expression also directly promotes tumor growth and invasion in a WNT, PI3K (phosphatidylinositol 3-kinase), and MAPK (mitogen-activated protein kinase)–dependent manner. Beyond primary cancers, AQP5 further enriches for a functional CSC population in metastatic tumors. Together, our findings support a CSC model in gastric tumors that may have application for therapeutic strategies targeting CSCs.
肿瘤驻留干细胞被认为有助于癌症进展和长期复发,但它们在胃癌中的身份和功能仍不清楚。Lim等人报告称,表面标志物AQP5在小鼠和人类源自胃幽门远端的肿瘤中富集了胃癌干细胞。AQP5阳性干细胞在移植后能够重建肿瘤,并生成长寿的类器官培养物。在实验模型系统中,选择性消融AQP5与肿瘤消退相关。干细胞上的AQP5表达调节了WNT、PI3K和MAPK信号传导,并在几种胃癌实验模型中增强了肿瘤增殖能力。—Priscilla N. Kelly
癌症干细胞是具有自我更新能力的群体,能够产生分化的肿瘤主体并维持这些癌症的长期存续。这些特性赋予癌症干细胞驱动持续肿瘤进展以及在癌症治疗后导致肿瘤复发的能力。定义并选择性消除癌症干细胞群体具有作为治疗策略的潜力。癌症干细胞已在血液、大脑和结肠等多种癌症中被鉴定出来,但迄今为止,对胃癌中癌症干细胞身份的稳健研究和验证仍然难以捉摸,阻碍了在此背景下对其作用的考察。
尽管具有生物学和治疗学意义,但在胃癌中鉴定稳健的癌症干细胞表面标志物仍面临几个主要障碍:(i)许多癌症干细胞标志物在细胞和组织类型中广泛表达,限制了它们用于分离这些群体的效用;(ii)大多数标志物的验证不足,依赖于标志物关联性,而非提供干细胞潜能读数的功能测定;(iii)许多模型在重现人类癌症中存在的分子和形态学特征方面缺乏足够的生理相关性。因此,需要制定一个统一的策略来鉴定胃癌中可能具有治疗潜力的癌症干细胞标志物。
我们开发了从小鼠和人类幽门部肿瘤组织(包括转移性肿瘤样本)中分离水通道蛋白5阳性癌症干细胞的方法。转录组分析显示,AQP5阳性肿瘤细胞富集于一个表达与干细胞活性相关基因的上皮细胞亚群中。我们通过多项功能测定证实了其癌症干细胞潜能,证明小鼠和人类的AQP5阳性细胞能够启动长寿的类器官培养,并在移植到小鼠体内时重建侵袭性肿瘤,而AQP5阴性细胞则无法做到。与这些发现一致,靶向消融AQP5阳性细胞阻断了肿瘤发生,并在已形成的肿瘤中驱动了肿瘤消退;通过重复消融策略可以消除这些肿瘤,以规避肿瘤可塑性。从功能角度来看,我们发现这些胃癌干细胞中的AQP5表达通过WNT、PI3K(磷脂酰肌醇3-激酶)和MAPK(丝裂原活化蛋白激酶)信号传导,在多样化的、接近生理条件的胃癌类器官和小鼠模型中促进了癌细胞的增殖和侵袭,这凸显了我们研究发现的更广泛相关性。
我们鉴定出了一个胃癌干细胞的表面标志物,该标志物在驱动癌症进展和转移中也发挥着功能性作用。与先前提出的标志物不同,AQP5能在小鼠和人类胃癌细胞的特定亚群中稳健地富集癌症干细胞,并在其中促进关键的癌症特性。这些发现凸显了在各种背景下(包括肿瘤可塑性、复发和治疗性耐药)研究癌症干细胞-微环境相互作用贡献的可能性,并可能有助于开发针对胃癌中AQP5阳性癌症干细胞的靶向疗法。
癌症干细胞(CSCs)是一类能够驱动长期肿瘤生长的自我更新群体。在胃癌中,CSCs群体的身份尚不明确。本研究建立了一个以水通道蛋白5(AQP5)为标志的胃癌干细胞群体,该群体存在于人类和小鼠的幽门部肿瘤中。通过多种类器官和小鼠模型,我们发现AQP5+ CSCs在癌症发生和持续进展中均不可或缺,并证明AQP5表达还通过WNT、PI3K(磷脂酰肌醇3-激酶)和MAPK(丝裂原活化蛋白激酶)依赖性方式直接促进肿瘤生长与侵袭。除了原发性癌症,AQP5在转移性肿瘤中进一步富集了功能性CSCs群体。综上,我们的研究结果支持胃癌中存在癌症干细胞模型,这或可为针对CSCs的治疗策略提供应用方向。
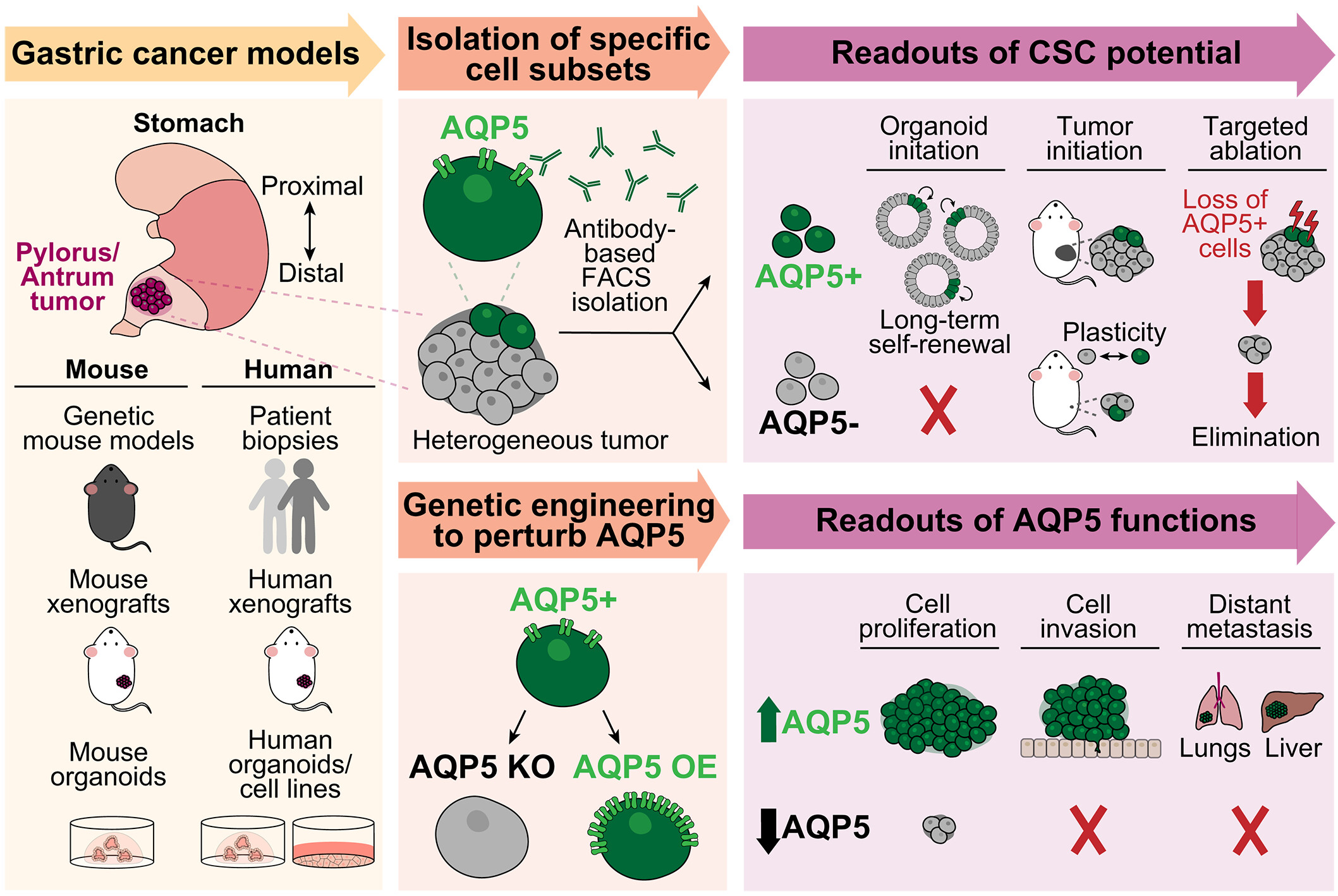
图:AQP5是小鼠和人类胃癌干细胞的功能标记物。我们建立了多种体外和体内胃癌模型。这些模型产生的肿瘤具有异质性,并包含AQP5阳性细胞亚群,该亚群可被选择性分离。多项实验表明AQP5阳性群体具有癌症干细胞潜能。此外,通过构建AQP5基因敲除和过表达细胞,我们揭示了AQP5在癌细胞增殖、侵袭和远处转移中的功能性作用。FACS,即荧光激活细胞分选技术。
AQP5: A functional gastric cancer stem cell marker in mouse and human tumors