Aging of the immune system is a key factor for cancer outcome. Park et al. performed immune profiling to explore how the immune system becomes less able to combat cancer with age. Through experimental lung cancer models, aging was associated with a more rapid recruitment of myeloid immune cells into tumors and enhanced tumor growth in older mice. Production of the cytokine interleukin-1α (IL-1α) by myeloid cells provided signals to hematopoietic progenitor cells in the bone marrow, which created a mechanism to further promote the recruitment of an immunosuppressive myeloid population. The enzyme DNMT3A (DNA methyltransferase 3A), involved in age-associated DNA methylation, was down-regulated with age and correlated with the production of IL-1. Blocking of the IL-1 receptor was found to reduce lung cancer progression in aged mice. —Priscilla N. Kelly
Most cancers are diseases of aging. Extensive epidemiological work has established that this association is most notable at the sixth decade of life in terms of frequency of cancer diagnoses. Aging is the number one correlate to an increased risk for developing cancer, lower overall and progression-free survival, and an increased likelihood for a future relapse or recurrence.
Studies on the cellular and molecular changes linked to aging have been attributed to the association between aging and cancer. And although preclinical studies testing therapeutics targeted toward senescent cells, exhausted T cells, or inflammatory molecules have shown some promise in patients, the field lacks a comprehensive understanding of whether tumor-intrinsic or -extrinsic and immunological or nonimmunological determinants of the tumor microenvironment are responsible for the protumorigenic consequence of aging. Recent work has demonstrated that cell-intrinsic aging of epithelial cells actually deters their neoplastic transformation and growth in old mice. Others have shown that, although a poor T cell response is associated with the protumor effect of aging, transferring T cells from young mice to old mice does not rescue the antitumor response in those recipients. As a result, there remains an unmet clinical need for an unbiased dissection of the tumor microenvironment that reveals how aging promotes cancer.
We show that the aging of the immune system, regardless of the age of the stroma, is associated with lung cancer. Notably, reconstituting old mice with a youthful immune system rescues their antitumor response and delays lung cancer progression. Hematopoietic aging enhances emergency myelopoiesis in old mice and results in an extramedullary accumulation of myeloid progenitors in lung tumors. Tumor-infiltrating, progenitor-like myeloid cells in old mice produce interleukin (IL)–1⍺, which acts as a driver of the enhanced myelopoietic response that promotes immunosuppression; disrupting this axis with either an anti–IL-1⍺ antibody or the IL-1 receptor 1 (IL-1R1) antagonist, anakinra, early during tumor initiation not only slowed tumor growth but also normalized myelopoiesis. The increased proclivity of aged monocyte-derived macrophages (mo-macs) to produce more IL-1 is due to an age-associated decline of DNA methyltransferase 3A (DNMT3A), the loss-of-function mutation of which is commonly linked to clonal hematopoiesis. Extending these findings to humans, we show that emergency hematopoiesis is enhanced with age and cancer stage; circulating hematopoietic progenitors from older patients exhibited a down-regulation of DNMT3A, and monocytes from older patients produced more IL-1. Moreover, in human lung cancer lesions, we show that enrichment for mo-macs that express IL-1⍺ correlates with aging, poorer survival, and recurrence of cancer.
We established that an aged immune system promotes tumor growth, regardless of the age of the tumor or its surrounding stroma. Specifically, hematopoietic aging drives emergency myelopoiesis, and targeting IL-1R1 signaling during early tumor development to attenuate this process abrogates the protumorigenic effect of aging on tumor control. Our study not only highlights the importance of immunotherapy in improving the age-dependent antitumor response but also the major role of local myeloid progenitors that actually fuel age-enhanced emergency myelopoiesis in a positive feedback loop. Notably, we defined the therapeutic window for the use of an IL-1R1–based intervention to delay lung cancer progression. Our findings have direct relevance to the design of cancer prevention strategies, support recent work from our group on lung cancer as a systemic disease involving the bone marrow, and are highly informative for unraveling the link between aging, DNMT3A mutation-driven clonal hematopoiesis, and cancer.
Age is a major risk factor for cancer, but how aging impacts tumor control remains unclear. In this study, we establish that aging of the immune system, regardless of the age of the stroma and tumor, drives lung cancer progression. Hematopoietic aging enhances emergency myelopoiesis, resulting in the local accumulation of myeloid progenitor–like cells in lung tumors. These cells are a major source of interleukin (IL)–1⍺, which drives the enhanced myeloid response. The age-associated decline of DNA methyltransferase 3A enhances IL-1⍺ production, and disrupting IL-1 receptor 1 signaling early during tumor development normalized myelopoiesis and slowed the growth of lung, colonic, and pancreatic tumors. In human tumors, we identified an enrichment for IL-1⍺–expressing monocyte-derived macrophages linked to age, poorer survival, and recurrence, unraveling how aging promotes cancer and offering actionable therapeutic strategies.
免疫系统的衰老是癌症预后的关键因素。Park等人进行了免疫分析,以探索免疫系统如何随年龄增长而变得更难对抗癌症。通过实验性肺癌模型,衰老与髓系免疫细胞更快速招募到肿瘤中以及老年小鼠肿瘤生长增强相关。髓系细胞产生的细胞因子白细胞介素-1α (IL-1α) 向骨髓中的造血祖细胞提供信号,这创建了一种机制,进一步促进免疫抑制性髓系群体的招募。与年龄相关DNA甲基化有关的酶DNMT3A (DNA甲基转移酶3A) 随年龄下调,并与IL-1的产生相关。阻断IL-1受体被发现可以减少老年小鼠的肺癌进展。—Priscilla N. Kelly
大多数癌症是衰老相关疾病。广泛的流行病学工作已确定,这种关联在癌症诊断频率方面在生命的第六个十年最为显著。衰老是增加患癌风险、降低总生存期和无进展生存期以及增加未来复发或再发可能性的首要相关因素。
与衰老相关的细胞和分子变化的研究被归因于衰老与癌症之间的关联。尽管针对衰老细胞、耗竭T细胞或炎症分子的治疗方法的临床前研究在患者中显示出一些希望,但该领域缺乏全面理解,即肿瘤微环境中的肿瘤内在或外在以及免疫或非免疫决定因素是否对衰老的促肿瘤后果负责。最近的工作表明,上皮细胞的细胞内在衰老实际上阻碍了它们在老年小鼠中的肿瘤转化和生长。其他人已经表明,尽管T细胞反应差与衰老的促肿瘤效应相关,但将年轻小鼠的T细胞转移到老年小鼠并不能挽救这些受体的抗肿瘤反应。因此,仍然存在未满足的临床需求,需要对肿瘤微环境进行无偏解剖,以揭示衰老如何促进癌症。
我们表明,无论基质年龄如何,免疫系统的衰老都与肺癌相关。值得注意的是,用年轻的免疫系统重建老年小鼠可以挽救它们的抗肿瘤反应并延缓肺癌进展。造血衰老增强了老年小鼠的应急髓系生成,导致髓系祖细胞样细胞在肺肿瘤中的髓外积累。老年小鼠中肿瘤浸润的祖细胞样髓系细胞产生白细胞介素 (IL)–1⍺,这作为增强的髓系生成反应的驱动因子,促进免疫抑制;在肿瘤发生早期使用抗–IL-1⍺抗体或IL-1受体1 (IL-1R1) 拮抗剂阿那白滞素破坏这一轴心,不仅减缓了肿瘤生长,而且使髓系生成正常化。老年单核细胞来源的巨噬细胞 (mo-macs) 产生更多IL-1的倾向增加是由于与年龄相关的DNA甲基转移酶3A (DNMT3A) 下降,其功能丧失突变通常与克隆性造血相关。将这些发现扩展到人类,我们表明应急造血随年龄和癌症阶段增强;老年患者的循环造血祖细胞表现出DNMT3A的下调,老年患者的单核细胞产生更多IL-1。此外,在人类肺癌病变中,我们表明表达IL-1⍺的mo-macs的富集与衰老、较差的生存率和癌症复发相关。
我们确定,无论肿瘤或其周围基质的年龄如何,衰老的免疫系统都会促进肿瘤生长。具体来说,造血衰老驱动应急髓系生成,在肿瘤发展早期靶向IL-1R1信号传导以减弱这一过程,消除了衰老对肿瘤控制的促肿瘤效应。我们的研究不仅强调了免疫疗法在改善年龄依赖性抗肿瘤反应中的重要性,而且强调了局部髓系祖细胞的主要作用,这些祖细胞实际上以正反馈循环推动年龄增强的应急髓系生成。值得注意的是,我们确定了使用基于IL-1R1的干预措施延缓肺癌进展的治疗窗口。我们的发现与癌症预防策略的设计直接相关,支持我们小组最近关于肺癌作为涉及骨髓的系统性疾病的工作,并对揭示衰老、DNMT3A突变驱动的克隆性造血和癌症之间的联系提供了重要信息。
年龄是癌症的主要风险因素,但衰老如何影响肿瘤控制仍不清楚。在这项研究中,我们确定,无论基质和肿瘤的年龄如何,免疫系统的衰老都驱动肺癌进展。造血衰老增强应急髓系生成,导致髓系祖细胞样细胞在肺肿瘤中的局部积累。这些细胞是白细胞介素 (IL)–1⍺的主要来源,驱动增强的髓系反应。与年龄相关的DNA甲基转移酶3A下降增强IL-1⍺产生,在肿瘤发展早期破坏IL-1受体1信号传导使髓系生成正常化并减缓肺、结肠和胰腺肿瘤的生长。在人类肿瘤中,我们发现了与年龄、较差的生存率和复发相关的表达IL-1⍺的单核细胞来源巨噬细胞的富集,揭示了衰老如何促进癌症并提供了可行的治疗策略。
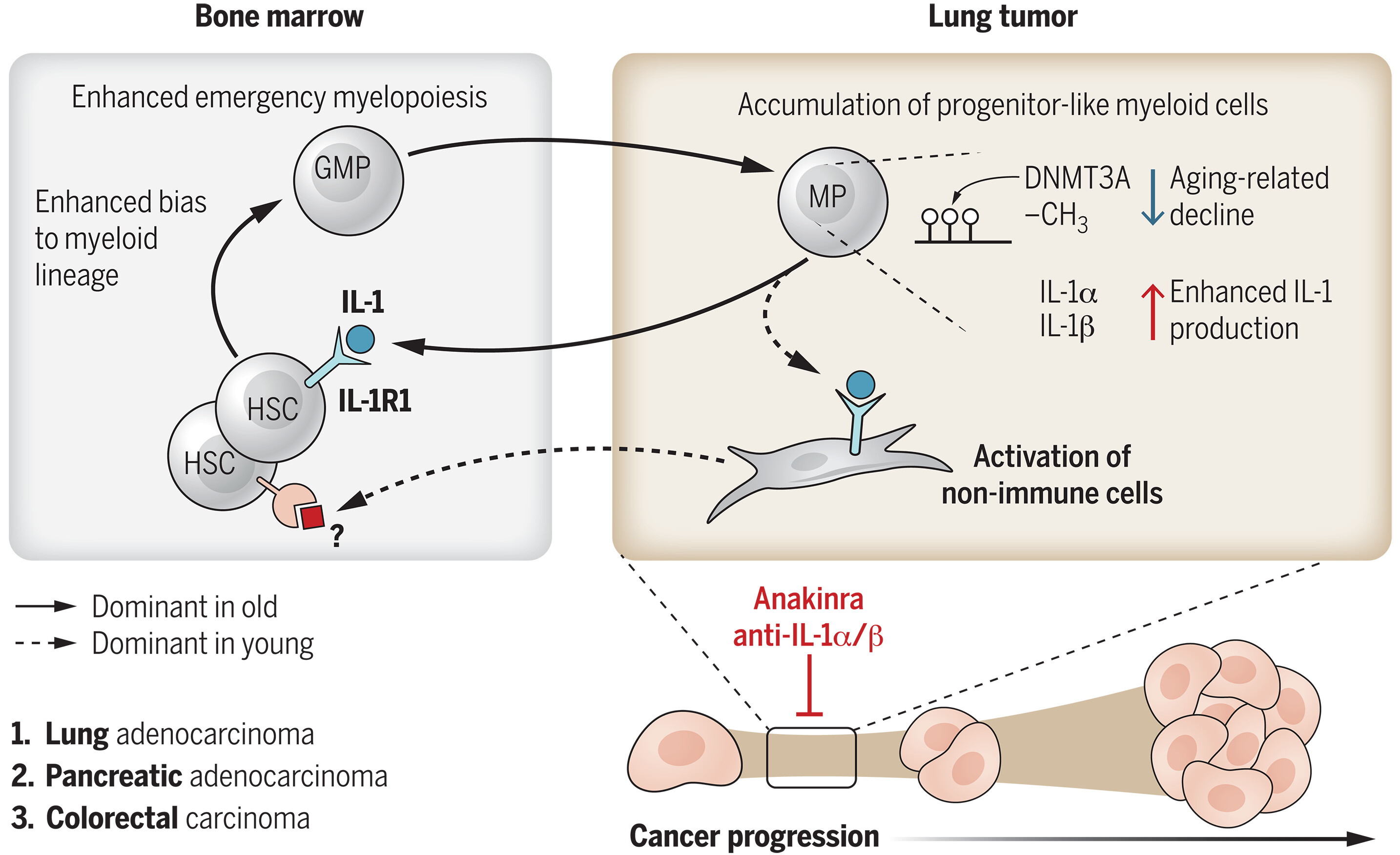
图:IL-1驱动的应急性髓系造血模型作为促肿瘤发生的造血衰老基础。免疫系统衰老通过加剧应急性髓系造血,并借助IL-1参与的正反馈循环维持该过程,从而促进肿瘤生长。在小鼠模型中,于癌症进展早期破坏该循环可延缓年龄依赖性癌症生长。GMP,粒细胞-单核细胞祖细胞;HSC,造血干细胞;MP,髓系祖细胞;CH3,甲基。
Hematopoietic aging promotes cancer by fueling IL-1⍺–driven emergency myelopoiesis