Cachexia, or wasting syndrome, arises during advanced stages of many chronic diseases, including cancer, and is often associated with loss of motivation and apathy. It is known that inflammation is a critical factor in cancer cachexia. However, the link between tumor-induced inflammation and the underlying brain mechanisms that produce motivational deficits remains elusive. Using a well-established mouse model of cancer cachexia, Zhu et al. uncovered a brainstem-to-basal ganglia circuit that suppresses dopamine signaling (see the Perspective by Dalley and Lynall). Neurons in the area postrema sense the inflammatory cytokine interleukin-6 and relay this signal to the parabrachial nucleus. The parabrachial nucleus drives inhibitory neurons in the substantia nigra pars reticulata, which then suppress dopamine neurons in the ventral tegmental area. This sequence reduces dopamine levels in the nucleus accumbens, ultimately increasing effort sensitivity and lowering motivation. —Peter Stern
Cachexia is a debilitating wasting syndrome that affects most advanced cancer patients, characterized by profound involuntary weight loss, muscle and fat depletion, and disrupted energy balance. Beyond physical decline, patients commonly experience severe fatigue, apathy, and depression that further diminish their quality of life. Despite these pervasive neuropsychiatric symptoms, the biological mechanisms linking peripheral wasting to brain dysfunction and behavioral changes remain poorly understood, hindering the development of effective treatments.
Systemic inflammation, marked by elevated circulating cytokines, is a key driver of cancer cachexia. Although inflammation could broadly compromise neuronal function, we hypothesized that it acts through specialized circuits that conserve energy during acute illness. In cancer, persistent activation of these circuits may drive chronic fatigue and depression, providing a mechanistic framework for how inflammation dynamically regulates motivation.
To investigate cachexia’s impact on motivation, we used an established preclinical model: mice subcutaneously implanted with colon adenocarcinoma cells (C26). Within weeks, the mice exhibited classical cachexia symptoms—weight loss, muscle wasting, and decreased feeding—accompanied by pronounced motivational deficits. A battery of behavioral assays uncovered specific deficits in effort-sensitive tasks such as patch foraging and progressive ratio tests. By contrast, tests of physical activity (open-field exploration), reward sensitivity (sucrose preference, a measure of anhedonia), and despair (tail suspension, forced swim tests) remained unchanged, although home-cage activity was reduced. This identifies a specific reduction in effort-based motivation, indicative of apathy and distinct from general weakness or anhedonia.
A comprehensive cytokine screen showed rising interleukin-6 (IL-6) levels in the blood and brain that paralleled cachexia progression. Whole-brain, cellular-resolution activity mapping and viral tracing uncovered a circuit originating in the area postrema, a circumventricular organ specialized for detecting blood-borne signals. IL-6–sensing neurons in this region project to the parabrachial nucleus, which in turn activates the substantia nigra pars reticulata inhibitory neurons, ultimately suppressing dopamine release in the nucleus accumbens. Optogenetic stimulation of area postrema neurons rapidly suppressed accumbens dopamine release. Longitudinal dopamine monitoring during cachexia progression revealed a progressive decline in accumbens dopamine that tracked with worsening motivational deficits.
Targeting this IL-6–sensing pathway reversed cachexia-induced apathy. Blocking IL-6 with systemic antibodies, knocking down (genetic reduction of gene expression) IL-6 receptors in the area postrema, or ablating area postrema–to–parabrachial neurons each alleviated the motivational deficits. Conversely, boosting dopamine signaling in the nucleus accumbens—through optogenetic stimulation of dopamine neurons or local dopamine agonist infusion—restored motivation despite ongoing inflammation, even at late stages of cancer progression.
We identified an immune-to-neural circuit through which inflammation drives motivational deficits in cancer cachexia. Area postrema neurons detect circulating IL-6 and relay this signal to the basal ganglia to suppress mesolimbic dopamine, thereby increasing effort sensitivity. This specialized interoceptive pathway demonstrates that inflammation engages discrete neural circuits rather than causing broad disruption and cumulative neural damage. Although this circuit is likely adaptive during acute illness—conserving energy by dampening motivation—its persistent engagement in chronic conditions such as cancer cachexia is detrimental. This reveals that psychiatric symptoms such as apathy in cachexia are not secondary effects of physical decline but can arise directly from the same pathological mechanisms driving the illness itself.
Targeting this circuit through ablation of IL-6 sensing or boosting dopamine release relieved motivational deficits, opening therapeutic avenues for neuropsychiatric symptoms in cachexia and possibly other inflammatory conditions where IL-6 is elevated. Moreover, by quantifying effort sensitivity as a dimension of motivation that is measurable across species establishes that inflammation specifically drives apathy-like behavior, enabling clinical translation and treatments for inflammation-driven motivational deficits common to conditions ranging from cancer to depression.
Cachexia, a severe wasting syndrome associated with inflammatory conditions, often leads to multiorgan failure and death. Patients with cachexia experience extreme fatigue, apathy, and clinical depression, yet the biological mechanisms underlying these behavioral symptoms and their relationship to the disease remain unclear. In a mouse cancer model, cachexia specifically induced increased effort-sensitivity, apathy-like symptoms through a cytokine-sensing brainstem-to-basal ganglia circuit. This neural circuit detects elevated interleukin-6 (IL-6) at cachexia onset and translates inflammatory signals into decreased mesolimbic dopamine, thereby increasing effort sensitivity. We alleviated these apathy-like symptoms by targeting key circuit nodes: administering an anti–IL-6 antibody treatment, ablating cytokine sensing in the brainstem, and optogenetically or pharmacologically boosting mesolimbic dopamine. Our findings uncovered a central neural circuit that senses systemic inflammation and orchestrates behavioral changes, providing mechanistic insights into the connection between chronic inflammation and depressive symptoms.
恶病质,或称消耗综合征,出现在包括癌症在内的许多慢性疾病的晚期阶段,并常与动力丧失和冷漠相关。众所周知,炎症是癌症恶病质的一个关键因素。然而,肿瘤诱导的炎症与产生动机缺陷的潜在大脑机制之间的联系仍然不明。使用一个成熟的癌症恶病质小鼠模型,朱等人揭示了一条抑制多巴胺信号传导的脑干到基底神经节回路(参见 Dalley 和 Lynall 的视角)。最后区的神经元感知炎症细胞因子白细胞介素-6,并将此信号传递给臂旁核。臂旁核驱动黑质网状部的抑制性神经元,进而抑制腹侧被盖区的多巴胺神经元。这一系列反应降低了伏隔核中的多巴胺水平,最终增加了努力敏感性并降低了动机。—Peter Stern
恶病质是一种使人衰弱的消耗综合征,影响大多数晚期癌症患者,其特征是严重的非自愿体重减轻、肌肉和脂肪消耗以及能量平衡失调。除了身体衰退,患者通常还会经历严重的疲劳、冷漠和抑郁,这进一步降低了他们的生活质量。尽管存在这些普遍存在的神经精神症状,但将外周消耗与大脑功能障碍和行为变化联系起来的生物学机制仍然知之甚少,这阻碍了有效疗法的开发。
全身性炎症,以循环细胞因子水平升高为标志,是癌症恶病质的关键驱动因素。虽然炎症可能广泛损害神经元功能,但我们假设它通过专门的在急性疾病期间保存能量的回路起作用。在癌症中,这些回路的持续激活可能导致慢性疲劳和抑郁,从而为炎症如何动态调节动机提供了一个机制框架。
为了研究恶病质对动机的影响,我们使用了一个成熟的临床前模型:皮下植入结肠腺癌细胞(C26)的小鼠。几周内,小鼠表现出典型的恶病质症状——体重减轻、肌肉消耗和进食减少——并伴有明显的动机缺陷。一系列行为分析揭示了在努力敏感性任务(如斑块觅食和渐进比率测试)中的特异性缺陷。相比之下,身体活动测试(旷场探索)、奖赏敏感性测试(蔗糖偏好,快感缺乏的测量)和绝望行为测试(悬尾、强迫游泳测试)则没有变化,尽管笼内活动减少。这确定了基于努力的动机特异性降低,表明是冷漠,而非一般性虚弱或快感缺乏。
全面的细胞因子筛查显示,血液和大脑中白细胞介素-6(IL-6)水平随着恶病质进展而升高。全脑细胞分辨率活动图谱和病毒示踪揭示了一条起始于最后区的回路,最后区是一个专门用于检测血源性信号的室周器官。该区域中感知IL-6的神经元投射到臂旁核,而臂旁核又激活黑质网状部的抑制性神经元,最终抑制伏隔核中的多巴胺释放。光遗传学刺激最后区神经元能快速抑制伏隔核的多巴胺释放。在恶病质进展过程中进行的纵向多巴胺监测显示,伏隔核多巴胺随着动机缺陷恶化而进行性下降。
靶向这一IL-6感知通路可以逆转恶病质诱导的冷漠。使用系统性抗体阻断IL-6、敲低(基因表达遗传性降低)最后区的IL-6受体或消融最后区到臂旁核的神经元,均能缓解动机缺陷。相反,尽管存在持续的炎症,甚至是在癌症进展的晚期阶段,通过光遗传学刺激多巴胺神经元或局部输注多巴胺激动剂来增强伏隔核的多巴胺信号,都能恢复动机。
我们发现了一条免疫到神经的回路,炎症通过该回路驱动癌症恶病质中的动机缺陷。最后区神经元检测循环中的IL-6,并将此信号传递给基底神经节以抑制中脑边缘多巴胺,从而增加努力敏感性。这条专门的内感受通路表明,炎症是通过作用于离散的神经回路,而不是引起广泛的破坏和累积的神经损伤。尽管该回路在急性疾病期间可能是适应性的——通过抑制动机来保存能量——但在癌症恶病质等慢性疾病中的持续激活则是有害的。这表明,恶病质中的冷漠等精神症状并非身体衰退的次要效应,而可以直接由驱动疾病本身的相同病理机制引起。
通过消融IL-6感知或增强多巴胺释放来靶向该回路,缓解了动机缺陷,这为恶病质以及可能其他IL-6升高的炎症性疾病中的神经精神症状开辟了治疗途径。此外,通过量化努力敏感性作为跨物种可测量的动机维度,确立了炎症特异性驱动冷漠样行为,这使得从癌症到抑郁症等一系列疾病中常见的炎症驱动性动机缺陷的临床转化和治疗成为可能。
恶病质是一种与炎症性疾病相关的严重消耗综合征,常导致多器官功能衰竭和死亡。恶病质患者会经历极度疲劳、冷漠和临床抑郁症,但这些行为症状背后的生物学机制及其与疾病的关系尚不清楚。在一个小鼠癌症模型中,恶病质通过一条细胞因子感知的脑干到基底神经节回路,特异性诱导了努力敏感性增加、冷漠样症状。该神经回路在恶病质发病时检测到升高的白细胞介素-6(IL-6),并将炎症信号转化为中脑边缘多巴胺的减少,从而增加努力敏感性。我们通过靶向关键回路节点缓解了这些冷漠样症状:施用抗IL-6抗体治疗、消融脑干中的细胞因子感知以及光遗传学或药理学增强中脑边缘多巴胺。我们的研究结果揭示了一条感知全身性炎症并协调行为变化的中枢神经回路,为慢性炎症与抑郁症状之间的联系提供了机制上的见解。
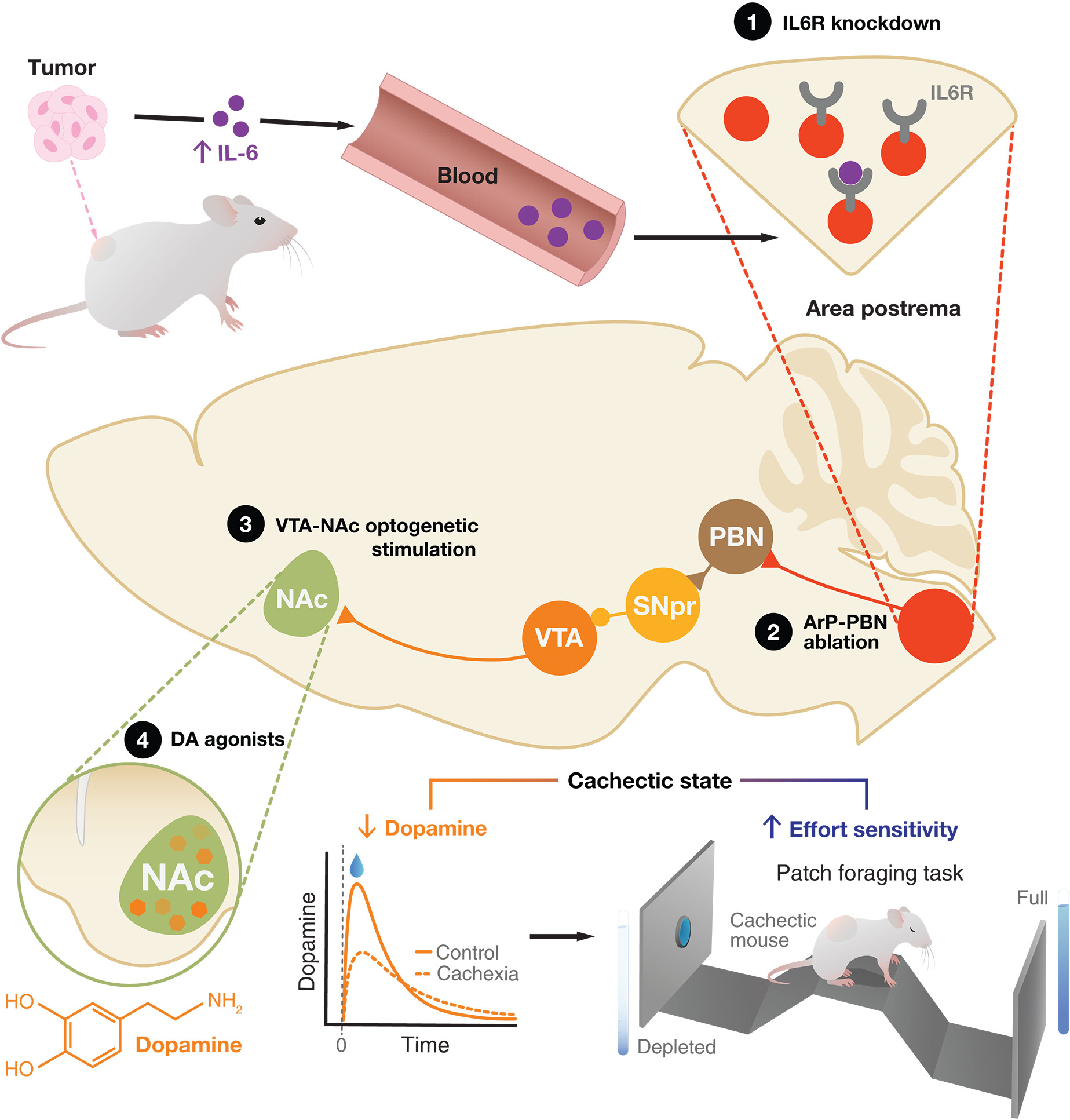
图:免疫-大脑环路驱动癌症恶病质中的冷漠样行为。最后区(ArP)在癌症恶病质中检测到升高的IL-6水平,导致伏隔核(NAc)中边缘中脑多巴胺减少。这种减少会增强努力敏感性,使恶病质小鼠产生冷漠样行为。通过功能增益或功能缺失操作靶向特定环路节点(黑色圆圈),可逆转这些行为。DA:多巴胺;PBN:臂旁核;SNpr:黑质网状部;VTA:腹侧被盖区。
A neuroimmune circuit mediates cancer cachexia-associated apathy