Cancer neoantigens are proteins expressed by tumor cells that can trigger an immune response that eliminates the tumor. A major challenge that limits their use as successful immunotherapy for solid tumors, particularly pancreatic cancer, is rarity of such cancer-specific proteins and peptides that are consistently expressed by most of the tumor cells. Ely et al. performed an immunopeptidomics study of human pancreatic cancers and identified a large number of unique peptides derived from noncoding regions of the genome (see the Perspective by Tuveson). Certain peptides were immunogenic and provided a source to generate T cell–specific immune responses that could kill pancreatic cancers in preclinical models. Some of the cancer-derived peptides were shared by multiple patients and were absent from normal pancreatic cells, raising the prospect of universal “off-the-shelf” therapies, as opposed to more challenging personalized approaches. —Priscilla N. Kelly
Cancer cells can translate ostensibly noncoding genetic elements to produce cryptic (noncanonical) peptides that can be presented by human leukocyte antigen class I (HLA-I). Cryptic peptides are poorly characterized in most solid tumors, where immunotherapies often yield limited efficacy. A deeper understanding of cryptic peptide presentation and immunogenicity could open new therapeutic avenues in solid tumors, such as pancreatic cancer.
Pancreatic cancer has been refractory to immunotherapies. A broader study of its antigen landscape, inclusive of noncanonical peptide sources, could identify antigens with potential cancer-restricted presentation. In this work, we investigated HLA-I–bound cryptic peptides in pancreatic cancer and assessed their immunogenic potential. We also developed a system to generate and functionally characterize T cell receptors (TCRs) that recognize these cryptic peptides.
We performed high-depth immunopeptidomics on organoids and bulk tumors from pancreatic cancer patients and found that organoids markedly improved specificity for cancer-derived peptides. Despite constructing personalized proteogenomic search spaces for each patient, we found that only a few mutation-encoded peptides were detectable using immunopeptidomics. By contrast, we empirically identified more than a thousand cryptic peptides presented by HLA-I. These cryptic peptides originated from diverse genomic elements, such as long noncoding RNAs (lncRNAs), 5′ or 3′ untranslated regions (UTRs), and alternative reading frames (intORFs).
We also evaluated a wide range of normal tissues, including healthy thymus, and nominated cryptic peptides with potential cancer restricted translation. Nearly 30% of cryptic peptides identified in pancreatic cancer were not detected in any healthy tissue profiled. We refer to these as cancer-restricted cryptic peptides. Many cancer-restricted cryptic peptides were shared by multiple patients, including nearly 50% of those predicted to bind to HLA-A*02:01.
We evaluated the immunogenic potential of cryptic peptides using an ex vivo T cell priming and expansion platform. Cancer-restricted cryptic peptides demonstrated robust immunogenicity, on par with that of mutation-encoded neoepitopes. Through barcode-enabled antigen mapping, we isolated TCRs specific for cryptic peptides at single-cell resolution. We reconstructed a subset of these TCRs to confirm antigen specificity, characterize TCR avidity, and assess potential cross-reactivity. Using CRISPR-based TCR redirection (TCR-T), we demonstrated that T cells could recognize endogenous levels of cryptic antigens on tumor cells. Furthermore, TCR-T cells specific to cancer-restricted cryptic antigens could exert robust killing of patient-derived pancreatic cancer organoids both ex vivo and in vivo.
We addressed two major gaps in the study of antigenic peptides in cancer—namely, the cancer restriction and immunogenicity of noncanonical peptides. These results demonstrate that aberrant translation in pancreatic cancer can give rise to cryptic antigens capable of recognition by T cells. Our data support that a subset of cryptic peptides are cancer restricted, immunogenic, and directly targetable on the surface of human pancreatic cancer cells, nominating them as potential therapeutic targets that warrant further investigation.
Translation of the noncoding genome in cancer can generate cryptic (noncanonical) peptides capable of presentation by human leukocyte antigen class I (HLA-I); however, the cancer specificity and immunogenicity of noncanonical HLA-I–bound peptides (ncHLAp) are incompletely understood. Using high-resolution immunopeptidomics, we discovered that cryptic peptides are abundant in the pancreatic cancer immunopeptidome. Approximately 30% of ncHLAp exhibited cancer-restricted translation, and a substantial subset were shared among patients. Cancer-restricted ncHLAp displayed robust immunogenic potential in a sensitive ex vivo T cell priming platform. ncHLAp-reactive, T cell receptor–redirected T cells exhibited tumoricidal activity against patient-derived pancreatic cancer organoids. These findings demonstrate that pancreatic cancer harbors cancer-restricted ncHLAp that can be recognized by cytotoxic T cells. Future therapeutic strategies for pancreatic cancer, and potentially other solid tumors, may include targeting cryptic antigens.
肿瘤新抗原是肿瘤细胞表达的蛋白质,可触发消除肿瘤的免疫反应。限制其作为实体瘤(尤其是胰腺癌)成功免疫疗法的一个主要挑战是,此类由大多数肿瘤细胞持续表达的癌症特异性蛋白质和肽段非常罕见。Ely等人对人类胰腺癌进行了免疫肽组学研究,鉴定了大量源自基因组非编码区域的独特肽段(参见Tuveson的展望)。某些肽段具有免疫原性,可作为产生T细胞特异性免疫反应的来源,在临床前模型中能够杀伤胰腺癌。部分源自癌症的肽段为多名患者所共有,且正常胰腺细胞中不存在,这提升了通用“现货型”疗法的前景,相较于更具挑战性的个性化方法而言。—Priscilla N. Kelly
癌细胞能够翻译表面上的非编码遗传元件,产生可被人白细胞抗原I类分子(HLA-I)呈递的隐性(非经典)肽段。在大多数实体瘤中,隐性肽段特征尚不明确,而这些肿瘤的免疫疗法往往效果有限。更深入地理解隐性肽段的呈递和免疫原性,可能为实体瘤(如胰腺癌)开辟新的治疗途径。
胰腺癌对免疫疗法反应不佳。对其抗原景观(包括非经典肽段来源)进行更广泛的研究,可能鉴定出具有潜在癌症限制性呈递的抗原。在本工作中,我们研究了胰腺癌中HLA-I结合的隐性肽段,并评估了其免疫原性潜力。我们还开发了一个系统来生成和功能性表征识别这些隐性肽段的T细胞受体(TCR)。
我们对胰腺癌患者的类器官和实体瘤进行了高深度免疫肽组学分析,发现类器官显著提高了对癌症来源肽段鉴定的特异性。尽管为每位患者构建了个性化的蛋白质基因组搜索空间,但我们发现仅能通过免疫肽组学检测到少数突变编码的肽段。相比之下,我们通过实验鉴定了一千多个由HLA-I呈递的隐性肽段。这些隐性肽段源自多种基因组元件,例如长链非编码RNA(lncRNA)、5‘或3’非翻译区(UTR)以及可变阅读框(intORF)。
我们还评估了包括健康胸腺在内的多种正常组织,并筛选出具有潜在癌症限制性翻译的隐性肽段。在胰腺癌中鉴定出的隐性肽段中,近30%在所分析的任何健康组织中均未检测到。我们称这些为癌症限制性隐性肽段。许多癌症限制性隐性肽段为多名患者所共有,包括预测可结合HLA-A*02:01的近50%的此类肽段。
我们使用离体T细胞启动和扩增平台评估了隐性肽段的免疫原性潜力。癌症限制性隐性肽段表现出强大的免疫原性,与突变编码的新表位相当。通过条形码编码的抗原定位技术,我们以单细胞分辨率分离了特异性识别隐性肽段的TCR。我们重构了这些TCR的一个子集,以确认抗原特异性、表征TCR亲和力并评估潜在的交叉反应性。利用基于CRISPR的TCR重定向技术(TCR-T),我们证明了T细胞能够识别肿瘤细胞上内源水平的隐性抗原。此外,特异性识别癌症限制性隐性抗原的TCR-T细胞能够在离体和体内对患者来源的胰腺癌类器官发挥强大的杀伤作用。
我们解决了癌症抗原肽研究中的两个主要空白——即非经典肽段的癌症限制性和免疫原性。这些结果表明,胰腺癌中的异常翻译能够产生可被T细胞识别的隐性抗原。我们的数据支持,一部分隐性肽段具有癌症限制性、免疫原性,并且能够在人类胰腺癌细胞表面被直接靶向,这将其提名为值得进一步研究的潜在治疗靶点。
癌症中非编码基因组的翻译能够产生可被人白细胞抗原I类分子(HLA-I)呈递的隐性(非经典)肽段;然而,人们对非经典HLA-I结合肽段(ncHLAp)的癌症特异性和免疫原性理解尚不完全。利用高分辨率免疫肽组学,我们发现隐性肽段在胰腺癌免疫肽组中含量丰富。大约30%的ncHLAp表现出癌症限制性翻译,并且相当一部分在患者间共享。癌症限制性ncHLAp在敏感的离体T细胞启动平台中显示出强大的免疫原性潜力。针对ncHLAp反应性的、经过T细胞受体重定向的T细胞对患者来源的胰腺癌类器官表现出杀瘤活性。这些发现表明,胰腺癌含有可被细胞毒性T细胞识别的癌症限制性ncHLAp。未来针对胰腺癌以及可能其他实体瘤的治疗策略,可能包括靶向隐性抗原。
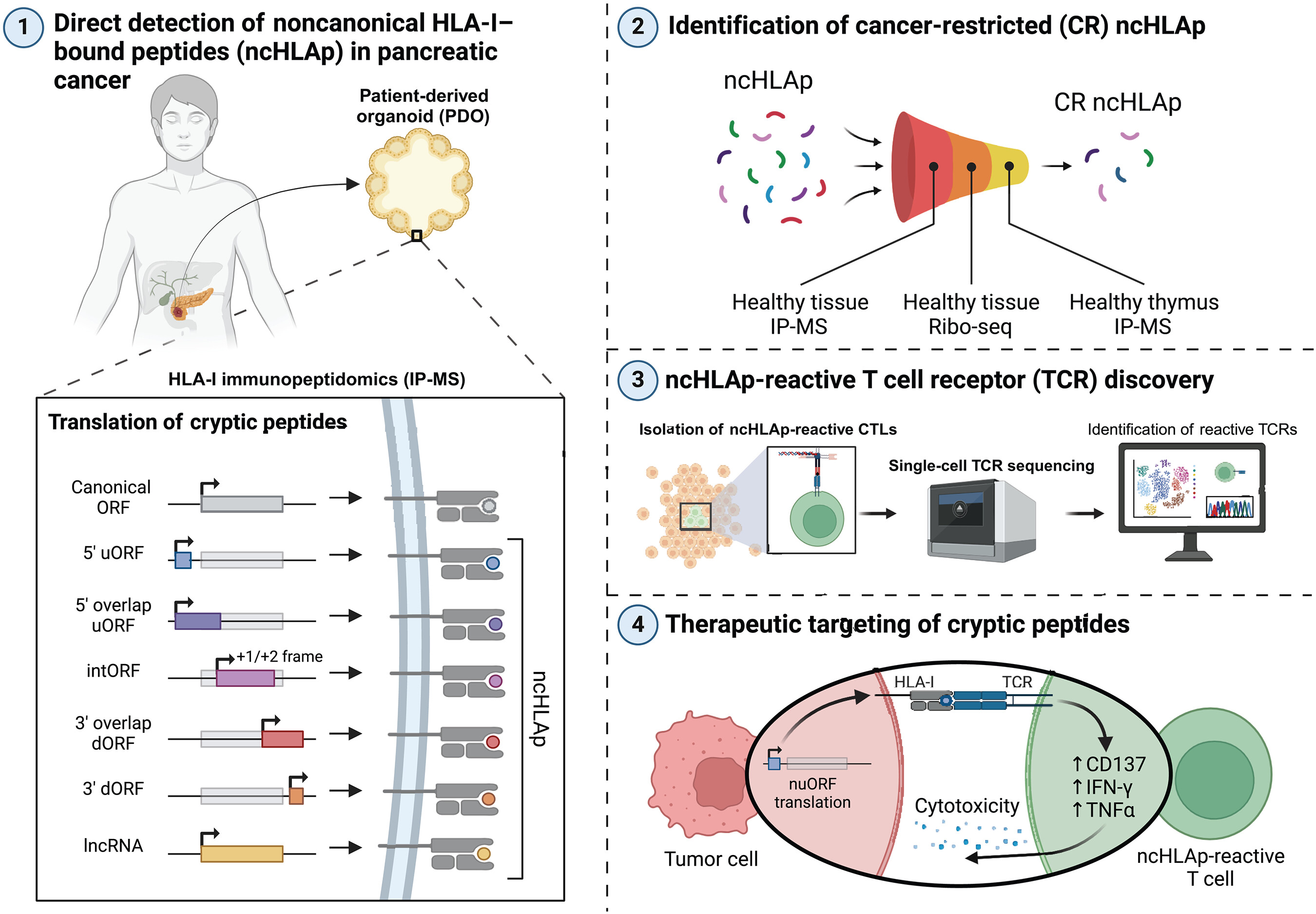
图:胰腺癌中的隐秘抗原。非编码基因的异常翻译会产生非常规(隐秘)肽段,这些肽段能够在胰腺癌中被 HLA-I 处理并呈递。隐秘肽段具有很强的免疫原性,相应的 TCR 能够在体外和体内识别并杀死患者来源的胰腺癌细胞团。nuORF,新未注释的开放阅读框;dORF,下游开放阅读框;Ribo-seq,核糖体测序;CTLs,细胞毒性 T 淋巴细胞;IFN-γ,干扰素-γ;TNFα,肿瘤坏死因子-α。
Pancreatic cancer–restricted cryptic antigens are targets for T cell recognition