B cells are white blood cells that produce antibodies and are often found within the tumor microenvironment. Ma et al. examined tumor-infiltrating B cells across 21 different cancer types from more than 270 patients (see the Perspective by Tellier and Nutt). The authors compiled single-cell transcriptome, B cell receptor repertoire, and chromatin accessibility data and report that tumor-associated B cells differentiated into antibody-secreting cells by either an extrafollicular pathway or by a more canonical germinal center pathway. Tumors associated with the extrafollicular B cell profile demonstrated poor clinical prognosis and resistance to immunotherapy compared with tumors harboring germinal center B cells. Alterations in the availability of glutamine-derived metabolites, which are known to influence T cell–dependent immunosuppression, may be linked to a dysfunctional humoral response and the adverse effect of extrafollicular B cells on tumors. —Priscilla N. Kelly
Tumor-infiltrating B cells have emerged as important players in cancer immunity and served as predictors of response to immunotherapy. These B cells display multiple functions, primarily through their ability to differentiate into plasma cells to produce antibodies, but vary spatiotemporally across different cancer types. Dissecting the abundance and differentiation states of B cells across diverse cancer types holds promise for improving the immunotherapeutic response.
To compile a comprehensive pan-cancer B cell landscape, we performed single-cell RNA sequencing (scRNA-seq) on paired tumors, lymph node metastases, adjacent normal tissues, and peripheral blood from patients with various cancer types, as well as incorporating substantial published scRNA-seq datasets. After correction of the batch effect, this atlas consists of scRNA-seq data from 269 patients across 20 cancer types. We assembled B cell receptor (BCR) sequencing of individual B cells with gene-expression profiles to characterize the dynamic transition between B cells and antibody-secreting cells (ASCs). We integrated the single-cell chromatin accessibility landscape of B cells from different cancers to dissect the epigenomic regulation networks that function in fine-tuning B cell development. We spatially localized B cells in mature versus immature tertiary lymphoid structures (TLSs) and investigated the potential regulators that direct B cells into specific responses.
We revealed substantial heterogeneity within B and plasma cells, identifying 15 B cell subsets and 10 plasma cell subsets. We computationally derived and validated two independent developmental pathways to ASCs through canonical germinal center (GC) and alternative extrafollicular (EF) pathways and demonstrated an apparent cancer-type preference. Colon adenocarcinoma and liver hepatocellular carcinoma were the two representative types of cancer enriched for GC and EF pathways, respectively. We affirmed that EF-dominant cancers correlate with dysregulated immune responses and worse clinical outcomes. We then identified the dynamic metabolic-epigenetic-signaling networks engaged in fine-tuning tumor-infiltrating B cell differentiation and managing the balance between the EF and GC pathways. Atypical memory (AtM) B cells, the primary progenitors of EF-derived ASCs, exhibit an exhausted and bystander phenotype and develop independently of the GC pathway. We found that the AtM B cells reside in the center of immature TLSs and spatially relocate to the periphery during TLS maturation. Last, we mechanistically linked these findings to specific transcription factors and epigenomic regulations. We demonstrated that the glutamine-derived metabolite α-ketoglutarate (α-KG) could increase the expression of AtM B cell–associated transcription factors T-bet and BATF and promote their differentiation, accompanied by the activation of mammalian target of rapamycin complex 1 (mTORC1) signaling. Consequently, AtM B cells acquire an immunoregulatory function that dampens antitumor T cell responses and fosters an immunosuppressive microenvironment.
We compiled the blueprint of B cell heterogeneity and two dynamic differentiation pathways in human cancers, providing a fundamental reference of ASC differentiation trajectory for future studies. The systematic comparison between EF and GC pathways reveals the similarities and differences of B cell states across different cancer types, highlighting the unfavorable clinical outcome linked to the immunosuppressive microenvironment of EF pathway–associated AtM B cells. Metabolic-epigenetic networks are remarkably flexible and can reconfigure B cell fates in a way that will facilitate the development of B cell–targeted immunotherapies.
B lymphocytes are essential mediators of humoral immunity and play multiple roles in human cancer. To decode the functions of tumor-infiltrating B cells, we generated a B cell blueprint encompassing single-cell transcriptome, B cell–receptor repertoire, and chromatin accessibility data across 20 different cancer types (477 samples, 269 patients). B cells harbored extraordinary heterogeneity and comprised 15 subsets, which could be grouped into two independent developmental paths (extrafollicular versus germinal center). Tumor types grouped into the extrafollicular pathway were linked with worse clinical outcomes and resistance to immunotherapy. The dysfunctional extrafollicular program was associated with glutamine-derived metabolites through epigenetic-metabolic cross-talk, which promoted a T cell–driven immunosuppressive program. These data suggest an intratumor B cell balance between extrafollicular and germinal-center responses and suggest that humoral immunity could possibly be harnessed for B cell–targeting immunotherapy.
B细胞是产生抗体的白细胞,常存在于肿瘤微环境中。Ma等人检测了来自270多名患者、21种不同癌症类型的肿瘤浸润B细胞(参见Tellier和Nutt的观点文章)。作者整合了单细胞转录组、B细胞受体库和染色质可及性数据,并报告指出,肿瘤相关B细胞通过滤泡外途径或更经典的生发中心途径分化为抗体分泌细胞。与含有生发中心B细胞的肿瘤相比,与滤泡外B细胞特征相关的肿瘤表现出较差的临床预后和对免疫疗法的抵抗性。已知能影响T细胞依赖性免疫抑制的谷氨酰胺衍生代谢物可用性的改变,可能与体液反应功能障碍以及滤泡外B细胞对肿瘤的不利影响有关。—Priscilla N. Kelly
肿瘤浸润B细胞已成为癌症免疫中的重要参与者,并可作为免疫疗法响应的预测指标。这些B细胞表现出多种功能,主要通过其分化为浆细胞产生抗体的能力,但在不同癌症类型中存在时空差异。剖析不同癌症类型中B细胞的丰度和分化状态,有望改善免疫治疗反应。
为了汇编一份全面的泛癌B细胞图谱,我们对来自不同癌症类型患者的配对肿瘤、淋巴结转移灶、癌旁正常组织和外周血进行了单细胞RNA测序(scRNA-seq),并整合了大量已发表的scRNA-seq数据集。经批次效应校正后,该图谱包含来自20种癌症类型、269名患者的scRNA-seq数据。我们将单个B细胞的B细胞受体(BCR)测序与基因表达谱整合,以表征B细胞与抗体分泌细胞(ASC)之间的动态转变。我们整合了来自不同癌症的B细胞的单细胞染色质可及性图谱,以剖析在精细调控B细胞发育中发挥功能的表观基因组调控网络。我们空间定位了成熟与未成熟三级淋巴结构(TLSs)中的B细胞,并研究了引导B细胞产生特异性反应的潜在调节因子。
我们揭示了B细胞和浆细胞内部的显著异质性,鉴定出15个B细胞亚群和10个浆细胞亚群。我们通过计算推导并验证了通过经典生发中心(GC)途径和替代性滤泡外(EF)途径分化为ASC的两个独立发育路径,并证明了明显的癌症类型偏好性。结肠腺癌和肝细胞癌分别是富含GC途径和EF途径的两种代表性癌症类型。我们证实,以EF为主的癌症与免疫反应失调和更差的临床结局相关。随后,我们鉴定了参与精细调控肿瘤浸润B细胞分化以及管理EF与GC途径之间平衡的动态代谢-表观遗传-信号网络。非典型记忆(AtM)B细胞是EF来源ASC的主要祖细胞,表现出耗竭和旁观者表型,并且独立于GC途径发育。我们发现AtM B细胞位于未成熟TLS的中心,并在TLS成熟过程中空间上重新定位到外周。最后,我们机制性地将这些发现与特定的转录因子和表观基因组调控联系起来。我们证明,谷氨酰胺衍生代谢物α-酮戊二酸(α-KG)可以增加AtM B细胞相关转录因子T-bet和BATF的表达并促进其分化,同时伴随哺乳动物雷帕霉素靶蛋白复合物1(mTORC1)信号通路的激活。因此,AtM B细胞获得了一种免疫调节功能,可抑制抗肿瘤T细胞反应并促进免疫抑制微环境。
我们汇编了人类癌症中B细胞异质性和两条动态分化路径的蓝图,为未来研究提供了ASC分化轨迹的基础参考。对EF和GC途径的系统性比较揭示了不同癌症类型间B细胞状态的异同,突出了与EF途径相关的AtM B细胞的免疫抑制微环境相关联的不利临床结局。代谢-表观遗传网络具有显著的灵活性,可以重新配置B细胞的命运,这将有助于开发靶向B细胞的免疫疗法。
B淋巴细胞是体液免疫的关键介质,在人类癌症中扮演多重角色。为了解码肿瘤浸润B细胞的功能,我们生成了一个B细胞蓝图,涵盖20种不同癌症类型(477个样本,269名患者)的单细胞转录组、B细胞受体库和染色质可及性数据。B细胞具有极大的异质性,包含15个亚群,可分为两条独立的发育路径(滤泡外途径与生发中心途径)。归类于滤泡外途径的肿瘤类型与较差的临床结局和对免疫疗法的抵抗性相关。功能失调的滤泡外程序通过表观遗传-代谢的交互对话与谷氨酰胺衍生代谢物相关联,从而促进了T细胞驱动的免疫抑制程序。这些数据提示了肿瘤内B细胞在滤泡外反应和生发中心反应之间存在平衡,并表明体液免疫可能被用于开发靶向B细胞的免疫疗法。
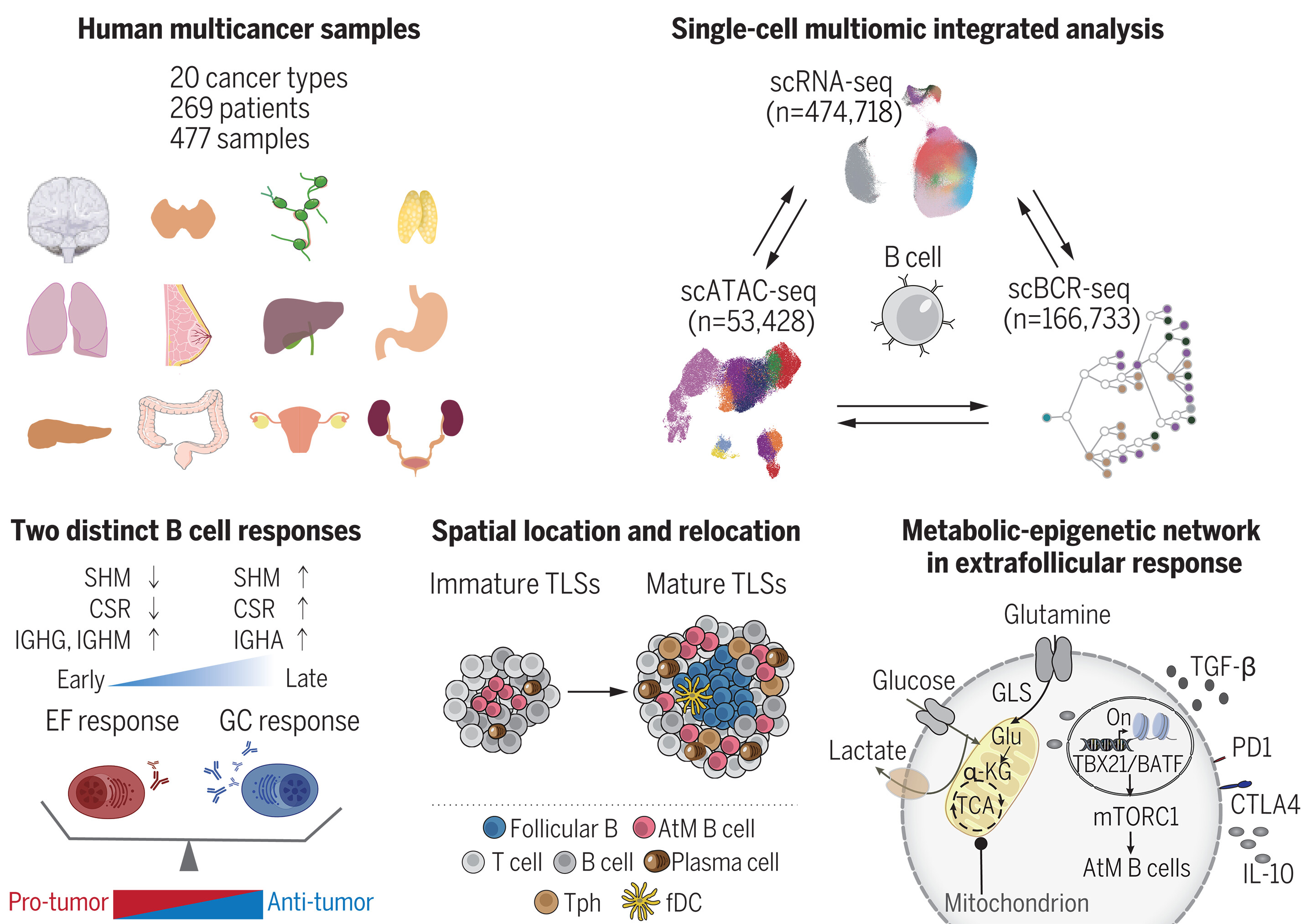
图:系统分析人类泛癌B细胞图谱。通过整合单细胞测序数据,我们对来自20种癌症类型269名患者的474,718个B细胞进行了分析。结合基因表达谱、B细胞受体序列及染色质可及性数据,我们深入探究了肿瘤浸润B细胞的多样性及可塑性,并在不同癌症类型间对EF反应性与GC反应性浆细胞进行了多层级比较。研究可视化展示了这些细胞沿三级淋巴结构成熟过程的动态空间定位,并揭示了调控B细胞分化的潜在代谢-表观遗传机制。
Germline-mediated immunoediting sculpts breast cancer subtypes and metastatic proclivity