Human leukocyte antigens (HLAs) are cell surface molecules that recognize antigenic peptides and present them to T cells to initiate an immune response. Krishna et al. performed an immunogenetic analysis of associations between HLA class II loci and lung cancer risk. They analyzed two large population-level datasets, one from the UK Biobank and the other from Finland’s FinnGen. Heterozygosity at HLA class II loci was found to be associated with reduced risk of developing lung cancer. Individuals who were current or former smokers benefited from this protective effect, but individuals who had never smoked did not. Smokers with lung cancer were found to have somatic loss of HLA class II heterozygosity, suggesting a functional role for HLA-related immune surveillance in cancer risk. —Priscilla N. Kelly
Whether the host immune system naturally protects against cancer has long been the subject of intense debate. The cancer immunosurveillance theory ascribes a protective function to the adaptive immune system, whereby T cell–mediated recognition of neoantigens presented by the major histocompatibility complex suppresses early neoplasia. Studies in mice have provided support for the cancer immunosurveillance theory, yet evidence for a protective role of the immune system against cancer in humans has been relatively lacking. In lung cancer, genetic variation in the human leukocyte antigen (HLA) locus is linked to tumor evolution and treatment outcomes, but whether HLA polymorphisms reduce lung cancer risk—which would imply a role of the host immune system in preventing lung cancer—is currently unclear. Population-scale biobank analysis coupling host genetics with longitudinal clinical data enables a systematic investigation of how HLA polymorphism influences lung cancer risk together with smoking and other established risk factors.
Understanding the molecular determinants of cancer risk is critical for early cancer detection and strategies to limit cancer mortality. Tobacco smoking increases lung cancer risk and is associated with a heightened somatic mutation rate that drives neoplastic potential, but whether there are additional risk factors that further modify lung cancer susceptibility, even among smokers, is unclear. The HLA heterozygote advantage theory posits that an HLA genotype encoding two different allomorphs enables presentation of a more diverse antigenic peptide repertoire to the immune system—and subsequent superior immune control of infected or cancerous cells—than does an HLA genotype encoding two equivalent allomorphs. Thus, heterozygous HLA allomorphs may present more neoantigens arising from smoking-derived somatic mutations. In this study, we evaluated the effect of HLA heterozygosity on lung cancer risk, leveraging genetic and longitudinal clinical data from the UK Biobank and FinnGen together with multimodal genomic analyses of nonmalignant and lung tumor samples.
In both the UK Biobank and FinnGen, we found that heterozygosity at the HLA class II (HLA-II) loci was associated with reduced risk of lung cancer over more than a decade of follow-up. HLA-II heterozygosity was associated with reduced risk of lung cancer in both current and former but not never-smokers, suggesting that smoking-derived antigens may augment the immune response to early neoplastic disease. HLA-II homozygosity conferred substantial lifetime risk of disease (e.g., in the UK Biobank, 13.9% for current smokers homozygous at HLA-DRB1) and was independent of known clinical and genetic risk factors, including a genome-wide polygenic risk score. Heterozygosity of amino acid sites within the HLA-II peptide binding groove was also associated with reduced risk of lung cancer, whereas analysis of single-cell RNA-sequencing data from nonmalignant and tumor lung samples showed that lung macrophages and epithelial cells express HLA-II and are affected by smoking. Analysis of tumor genomes from the The Cancer Genome Atlas (TCGA) cohort, the Pan-Cancer Analysis of Whole Genomes (PCAWG) cohort, and Hartwig Medical Foundation cohort revealed widespread loss of heterozygosity (LOH) of the HLA-II loci in lung cancer, with rates of LOH equaling those of HLA-I. An analysis of neoantigen repertoires between lung cancer tumors with and without HLA-II LOH showed that HLA-II LOH favors the loss of alleles with larger neopeptide repertoires, underscoring the importance of the HLA-II loci and the CD4+ T cell response in lung cancer.
The association of HLA-II heterozygosity with reduced risk of lung cancer implies that genetic variation in immunosurveillance is a feature of cancer susceptibility, together with environmental exposures, hereditary risk, and DNA replication errors. Our findings broaden understanding of the role of the host immune system in cancer risk and may motivate the incorporation of immunogenetics into lung cancer screening programs.
Cancer risk is influenced by inherited mutations, DNA replication errors, and environmental factors. However, the influence of genetic variation in immunosurveillance on cancer risk is not well understood. Leveraging population-level data from the UK Biobank and FinnGen, we show that heterozygosity at the human leukocyte antigen (HLA)-II loci is associated with reduced lung cancer risk in smokers. Fine-mapping implicated amino acid heterozygosity in the HLA-II peptide binding groove in reduced lung cancer risk, and single-cell analyses showed that smoking drives enrichment of proinflammatory lung macrophages and HLA-II+ epithelial cells. In lung cancer, widespread loss of HLA-II heterozygosity (LOH) favored loss of alleles with larger neopeptide repertoires. Thus, our findings nominate genetic variation in immunosurveillance as a critical risk factor for lung cancer.
人类白细胞抗原(HLA)是细胞表面分子,能够识别抗原肽并将其呈递给T细胞以启动免疫应答。Krishna等人对HLA II类基因座与肺癌风险之间的关联进行了免疫遗传学分析。他们分析了两个大规模人群水平数据集,一个来自英国生物样本库(UK Biobank),另一个来自芬兰的FinnGen。研究发现,HLA II类基因座的杂合性与降低罹患肺癌的风险相关。当前或曾经吸烟的个体受益于这种保护效应,但从未吸烟的个体则没有。研究发现,患有肺癌的吸烟者存在HLA II类杂合性的体细胞丢失,这提示HLA相关的免疫监视在癌症风险中发挥功能性作用。—Priscilla N. Kelly
宿主的免疫系统是否能天然地保护机体免于癌症,长期以来一直是激烈争论的主题。癌症免疫监视理论将保护功能归因于适应性免疫系统,即T细胞介导的对主要组织相容性复合体呈递的新抗原的识别,从而抑制早期肿瘤形成。小鼠研究为癌症免疫监视理论提供了支持,然而关于人类免疫系统对癌症具有保护作用的证据相对缺乏。在肺癌中,人类白细胞抗原(HLA)基因座的遗传变异与肿瘤演变和治疗结果相关,但HLA多态性是否能降低肺癌风险——这将意味着宿主免疫系统在预防肺癌中的作用——目前尚不清楚。结合宿主遗传学和纵向临床数据的人群规模生物样本库分析,使得能够系统地研究HLA多态性如何与吸烟及其他已知风险因素共同影响肺癌风险。
理解癌症风险的分子决定因素对于早期癌症检测和降低癌症死亡率的策略至关重要。吸烟会增加肺癌风险,并与驱动肿瘤潜能的体细胞突变率升高相关,但是否存在进一步改变肺癌易感性的额外风险因素,甚至在吸烟者中,尚不清楚。HLA杂合优势理论认为,与编码两个相同同种异型的HLA基因型相比,编码两个不同同种异型的HLA基因型能够向免疫系统呈递更多样化的抗原肽库,从而对受感染或癌变细胞产生更优的免疫控制。因此,杂合的HLA同种异型可能呈递更多由吸烟衍生的体细胞突变产生的新抗原。在本研究中,我们利用英国生物样本库和FinnGen的遗传及纵向临床数据,并结合非恶性和肺肿瘤样本的多组学基因组分析,评估了HLA杂合性对肺癌风险的影响。
在英国生物样本库和FinnGen中,我们都发现,在超过十年的随访期间,HLA II类(HLA-II)基因座的杂合性与肺癌风险降低相关。HLA-II杂合性与当前吸烟者和既往吸烟者(而非从不吸烟者)的肺癌风险降低相关,这表明吸烟衍生的抗原可能增强了针对早期肿瘤疾病的免疫应答。HLA-II纯合性带来了显著的终生疾病风险(例如,在英国生物样本库中,HLA-DRB1纯合子的当前吸烟者风险为13.9%),且独立于已知的临床和遗传风险因素,包括全基因组多基因风险评分。HLA-II肽结合沟内的氨基酸位点杂合性也与肺癌风险降低相关,而对来自非恶性和肺肿瘤样本的单细胞RNA测序数据分析显示,肺巨噬细胞和上皮细胞表达HLA-II并受吸烟影响。对来自癌症基因组图谱(TCGA)队列、全基因组泛癌分析(PCAWG)队列和Hartwig医学基金会队列的肿瘤基因组分析揭示了肺癌中HLA-II基因座广泛的杂合性缺失(LOH),其LOH发生率与HLA-I相当。对存在和不存在HLA-II LOH的肺癌肿瘤新抗原库的分析表明,HLA-II LOH倾向于丢失具有更大新肽库的等位基因,这凸显了HLA-II基因座和CD4+ T细胞应答在肺癌中的重要性。
HLA-II杂合性与肺癌风险降低的关联意味着,除了环境暴露、遗传风险和DNA复制错误之外,免疫监视的遗传变异也是癌症易感性的一个特征。我们的发现拓宽了对宿主免疫系统在癌症风险中作用的理解,并可能推动将免疫遗传学纳入肺癌筛查项目。
癌症风险受遗传突变、DNA复制错误和环境因素的影响。然而,免疫监视的遗传变异对癌症风险的影响尚不清楚。利用来自英国生物样本库和FinnGen的人群水平数据,我们表明人类白细胞抗原(HLA)-II基因座的杂合性与吸烟者肺癌风险降低相关。精细定位提示HLA-II肽结合沟内的氨基酸杂合性与肺癌风险降低相关,单细胞分析显示吸烟驱动促炎性肺巨噬细胞和HLA-II+上皮细胞的富集。在肺癌中,广泛的HLA-II杂合性缺失(LOH)倾向于丢失具有更大新肽库的等位基因。因此,我们的研究结果表明免疫监视的遗传变异是肺癌的一个关键风险因素。
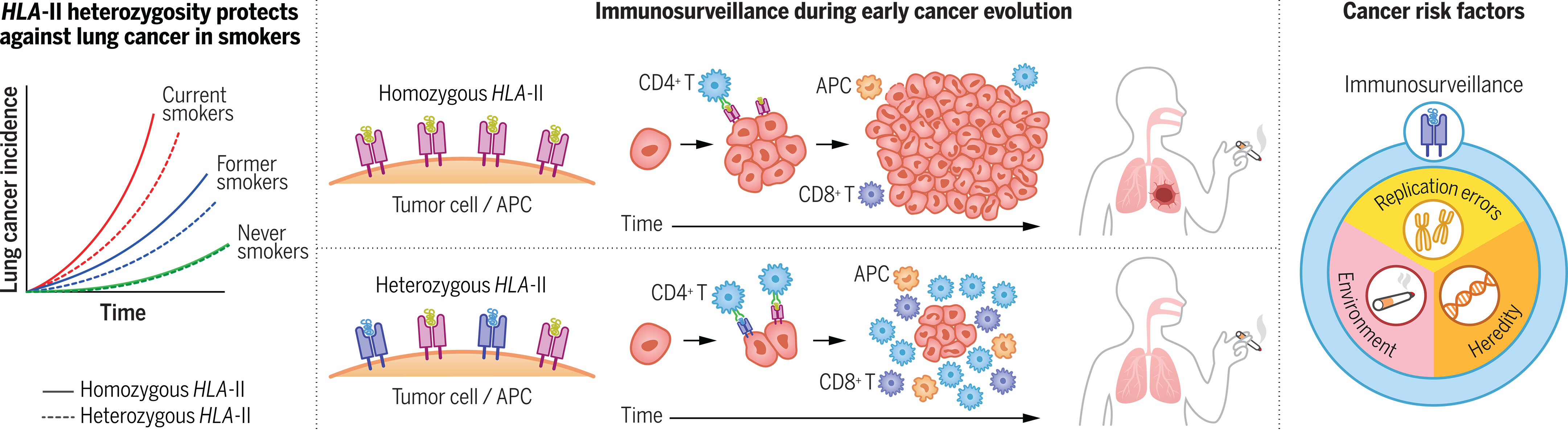
图:肺癌风险的免疫遗传学机制:基于英国生物样本库和FinnGen数据库的遗传流行病学分析,结合多组学研究发现,HLA-II杂合性能降低吸烟者的肺癌风险。数据显示,杂合性HLA同种异型可通过呈递多样化的吸烟相关抗原,增强机体对早期肿瘤的免疫监视能力。这表明免疫监视的遗传变异是癌症风险的关键影响因素。APC:抗原呈递细胞。