Cancers such as HER2-positive breast cancer are typically characterized by their location and marker proteins that influence how they behave and what treatments they respond to. However, not all cancers or tumors that nominally fall into a single category behave the same way. Some of these differences are caused by genetically encoded attributes affecting the tumor’s interactions with the immune system, as shown by Houlahan et al. (see the Perspective by Waddell and Addala). The authors demonstrated that a patient’s germline variants in an oncogene of interest, such as HER2, as well as their human leukocyte antigens responsible for the presentation of antigens to T cells, help to determine which tumors might survive and which ones will likely be destroyed by the patient’s immune system. —Yevgeniya Nusinovich
Cancer is defined by a set of abnormal biological capacities, termed the “hallmarks of cancer,” which can be acquired by hijacking various cellular processes. As a result, tumors with the same clinical characteristics can vary dramatically across individuals, and these distinct molecular vulnerabilities can have important prognostic and therapeutic implications. Oncogenic alterations are acquired within the context of an individual’s inherited DNA—known as the germline genome—which differ across individuals at millions of polymorphic sites. It remains poorly understood how inherited variants impact the evolution of a tumor. The most compelling example is that deleterious germline variants in BRCA1 and, to a lesser extent, BRCA2, are preferentially associated with the development of triple negative breast cancer and estrogen receptor (ER)–positive (ER+) breast cancer, respectively, implying that germline variants modulate specific subtypes of disease. The mechanistic basis for this preference is incompletely characterized. Various lines of evidence suggest that avoidance of the adaptive immune system determines which somatic mutations persist within a tumor. It remains less clear how germline differences influence immunoediting. Generally, germline variants have not been considered a source of immunogenic epitopes because cytotoxic response should be dampened by central and peripheral tolerance. However, nonmutated immunogenic epitopes have been identified in genes such as ERBB2 in breast and ovarian cancer and H4 histone in prostate cancer, among others, suggesting that germline variants may play a role in immunosurveillance.
We sought to investigate whether germline variants sculpt somatic evolution by mediating immunoediting. Specifically, we hypothesized that the burden of germline-derived epitopes in recurrently amplified driver genes may select against somatic gene amplification. This is because amplification of a gene with a high burden of germline-derived epitopes would increase epitope availability, likelihood of epitope presentation, and immune-mediated cell death. Thus, somatic amplification of the gene would come at a fitness cost in cells with a high burden of germline-derived epitopes. Instead, a tumor may favor an alternative oncogenic pathway with lower fitness costs.
We leveraged paired tumor and normal sequencing data from 4918 primary and 611 metastatic breast cancer patients, as well as somatic genomic profiles from 341 patients with ductal carcinoma in situ (DCIS), and evaluated the relationship between germline-derived epitope burden (GEB) and subtype commitment, defined by the acquisition of focal oncogenic amplifications. As proof of concept, we identified two immunogenic peptides derived from the germline sequence of human epidermal growth factor receptor 2 (HER2; peptides GP2 and E75) and hypothesized that the ability to present either peptide would be negatively associated with developing HER2+ breast cancer. We discovered that individuals who possess MHC class I alleles that can bind and present GP2 or E75 are less likely to develop HER2+ breast cancer than other breast cancer subtypes. In addition to GP2 and E75, we found that individuals with a high GEB in ERBB2, encoding HER2, are less likely to develop HER2+ breast cancer. The same negative association was observed for three recurrent amplicons observed in distinct ER+ breast cancers, which are characterized by high risk of relapse. Negative associations were robust to the definition of subtype, binding affinity threshold, and the algorithm to predict binding affinity. Tumors that overcame immune-mediated negative selection were more aggressive and exhibited microenvironments depleted of lymphocytes. In DCIS, before immune escape, high GEB was negatively associated with invasive breast cancer recurrence, suggesting that GEB is protective in the preinvasive setting.
These data indicate that supposedly “benign” germline variants with little to no functional genic effect may, in aggregate, sculpt breast cancer subtypes and disease aggression through immunoediting. These data also indicate that immunoediting pressures differ during the course of a patient’s disease, potentially informing the timing of therapeutic interventions. Exploiting germline-mediated immunoediting may inform the development of biomarkers that predict risk of invasive breast cancer recurrence and further refine risk stratification within invasive breast cancer subtypes.
Tumors with the same diagnosis can have different molecular profiles and response to treatment. It remains unclear when and why these differences arise. Somatic genomic aberrations occur within the context of a highly variable germline genome. Interrogating 5870 breast cancer lesions, we demonstrated that germline-derived epitopes in recurrently amplified genes influence somatic evolution by mediating immunoediting. Individuals with a high germline-epitope burden in human epidermal growth factor receptor 2 (HER2/ERBB2) are less likely to develop HER2-positive breast cancer compared with other subtypes. The same holds true for recurrent amplicons defining three aggressive estrogen receptor (ER)–positive subgroups. Tumors that overcome such immune-mediated negative selection are more aggressive and demonstrate an “immune cold” phenotype. These data show that the germline genome plays a role in dictating somatic evolution.
诸如HER2阳性乳腺癌等癌症通常根据其位置和标志蛋白来定义,这些蛋白影响其行为方式以及对治疗的反应。然而,并非所有名义上属于同一类别的癌症或肿瘤都以相同方式表现。Houlahan等人(参见Waddell和Addala的观点文章)表明,其中一些差异是由遗传编码的属性引起的,这些属性影响了肿瘤与免疫系统的相互作用。作者证明,患者特定癌基因(如HER2)中的胚系变异,以及负责向T细胞呈递抗原的人类白细胞抗原,共同决定了哪些肿瘤可能存活,哪些肿瘤很可能被患者的免疫系统摧毁。——Yevgeniya Nusinovich
癌症由一组异常的生物学能力定义,称为“癌症特征”,这些能力可通过劫持各种细胞过程获得。因此,具有相同临床特征的肿瘤在不同个体间差异巨大,而这些不同的分子脆弱性可能具有重要的预后和治疗意义。致癌性改变是在个体遗传DNA(称为胚系基因组)的背景下获得的,这些DNA在数百万个多态性位点上因人而异。目前对于遗传变异如何影响肿瘤进化仍知之甚少。最具说服力的例子是,BRCA1(以及程度较轻的BRCA2)的有害胚系变异分别优先与三阴性乳腺癌和雌激素受体(ER)阳性(ER+)乳腺癌的发生相关,这意味着胚系变异调节特定的疾病亚型。这种偏好性的机制基础尚未完全阐明。多方面证据表明,避开适应性免疫系统决定了哪些体细胞突变能在肿瘤中持续存在。胚系差异如何影响免疫编辑则尚不清楚。通常,胚系变异未被视为免疫原性表位的来源,因为细胞毒性反应应受到中枢和外周免疫耐受的抑制。然而,在乳腺癌和卵巢癌的ERBB2基因以及前列腺癌的H4组蛋白基因等中,已发现非突变的免疫原性表位,这提示胚系变异可能在免疫监视中发挥作用。
我们试图研究胚系变异是否通过介导免疫编辑来塑造体细胞进化。具体来说,我们假设,在反复扩增的驱动基因中,胚系来源表位的负荷可能不利于体细胞基因扩增。这是因为,对一个具有高胚系来源表位负荷的基因进行扩增,会增加表位的可用性、表位呈递的可能性以及免疫介导的细胞死亡。因此,在具有高胚系来源表位负荷的细胞中,对该基因进行体细胞扩增会带来适应度代价。相反,肿瘤可能倾向于选择具有较低适应度代价的替代致癌途径。
我们利用来自4918名原发性乳腺癌和611名转移性乳腺癌患者的配对肿瘤与正常组织测序数据,以及341名导管原位癌(DCIS)患者的体细胞基因组图谱,评估了胚系来源表位负荷(GEB)与亚型定向(通过获得局灶性致癌扩增来定义)之间的关系。作为概念验证,我们鉴定了两个源自人表皮生长因子受体2(HER2;肽段GP2和E75)胚系序列的免疫原性肽段,并假设能够呈递任一肽段的能力与发生HER2+乳腺癌呈负相关。我们发现,拥有能够结合并呈递GP2或E75的MHC I类等位基因的个体,相较于其他乳腺癌亚型,患HER2+乳腺癌的可能性更低。除了GP2和E75,我们还发现,在编码HER2的ERBB2基因中具有高GEB的个体,患HER2+乳腺癌的可能性更低。在不同ER+乳腺癌中观察到的、以高复发风险为特征的三种反复出现的扩增子也观察到了同样的负相关。这种负相关性对于亚型的定义、结合亲和力阈值以及预测结合亲和力的算法都是稳健的。那些克服了免疫介导负向选择的肿瘤更具侵袭性,并表现出淋巴细胞耗竭的微环境。在DCIS中,在免疫逃逸发生之前,高GEB与浸润性乳腺癌复发呈负相关,这表明GEB在浸润前阶段具有保护作用。
这些数据表明,那些被认为“良性”、几乎没有或完全没有功能性基因效应的胚系变异,可能总体上通过免疫编辑塑造了乳腺癌亚型和疾病的侵袭性。这些数据还表明,免疫编辑的压力在患者疾病过程中有所不同,这可能为治疗干预的时机提供参考。利用胚系介导的免疫编辑可能有助于开发生物标志物,以预测浸润性乳腺癌复发风险,并进一步细化浸润性乳腺癌亚型内部的风险分层。
相同诊断的肿瘤可能具有不同的分子谱和治疗反应。目前尚不清楚这些差异何时以及为何产生。体细胞基因组畸变发生在一个高度变异的胚系基因组背景下。通过对5870个乳腺癌病灶的分析,我们证明反复扩增基因中的胚系来源表位通过介导免疫编辑影响体细胞进化。与其它亚型相比,在人表皮生长因子受体2(HER2/ERBB2)中具有高胚系表位负荷的个体,患HER2阳性乳腺癌的可能性更低。对于定义了三个侵袭性雌激素受体(ER)阳性亚组的反复出现的扩增子,情况也是如此。那些克服了这种免疫介导负向选择的肿瘤更具侵袭性,并表现出“免疫冷”表型。这些数据表明,胚系基因组在决定体细胞进化方面发挥着作用。
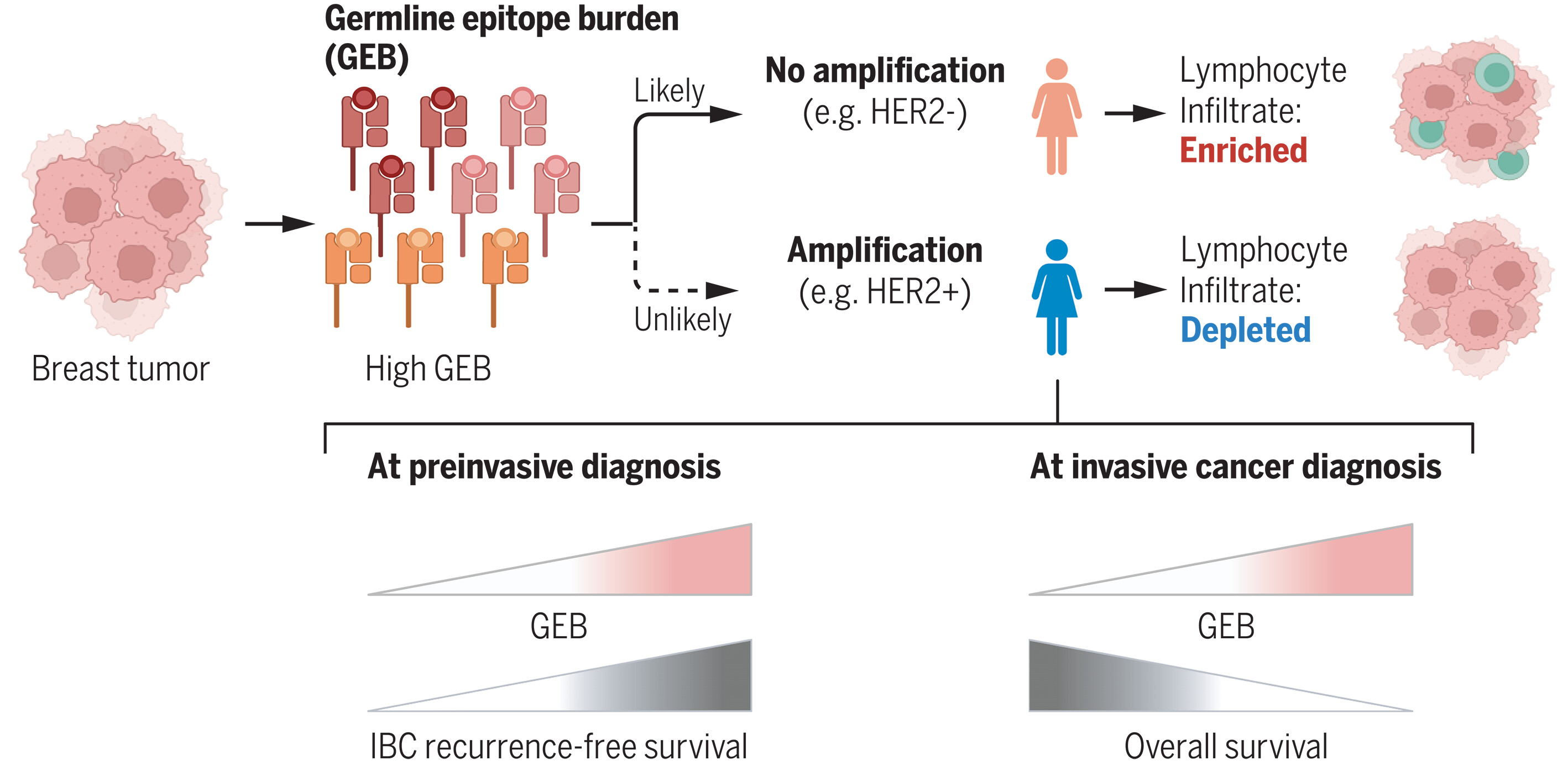
图:生殖系介导的免疫编辑塑造了癌症亚型与转移倾向性。在肿瘤发生过程中,癌症基因中生殖系基因高表达(GEB)水平较高的病变部位较难获得该基因的体细胞扩增。在浸润前乳腺肿瘤中,高GEB对浸润性乳腺癌的复发具有保护作用。然而,已形成的肿瘤若获得体细胞扩增,则被迫形成免疫抑制与免疫逃逸表型,侵袭性更强[图表由Biorender.com制作]。
Germline-mediated immunoediting sculpts breast cancer subtypes and metastatic proclivity