Treatment outcomes for breast cancer metastasis to the leptomeninges (LM), the cerebrospinal fluid–containing membranes that line the surface of the brain and spinal cord, are extremely poor. Whiteley et al. used experimental models of breast cancer LM metastasis to show that breast cancers expressing the cell surface receptor integrin α6 can invade the LM by traversing the outer surface of the blood vessels that normally connect the adjacent vertebral and skull bone marrow to the central nervous system meninges (see the Perspective by Monteran and Erez). Breast cancer cells then stimulate resident meningeal macrophages to secrete a neuronal prosurvival protein called glial-derived neurotrophic factor that supports tumor growth. These findings shed new light on tumor-host interactions in the LM and may guide new therapeutic targets for the treatment of LM metastasis. —Priscilla N. Kelly
Our group previously discovered that hematopoietic-lineage cells can traffic from the vertebral or skull bone marrow (BM) to the leptomeninges (LM) by migrating along the laminin-rich external surface of emissary veins (EVs). These EVs, part of the vertebral-calvarial BM vasculature, pass through foramina in the bone surface and emerge as LM blood vessels, thereby directly connecting the BM compartment to the central nervous system (CNS). This abluminal vascular migration route bypasses the need for cells to enter the circulation, cross the CNS blood-brain or blood–cerebrospinal fluid (CSF) barriers, or disrupt tissue boundaries.
The BM is the most frequent site of breast cancer (BC) metastasis, and most BC patients diagnosed with leptomeningeal disease (LMD) have established vertebral bone metastases. This observation suggests that EV trafficking could be an efficient mechanism of entry for BC cells (BCCs) into the LM; however, EV trafficking by carcinoma cells has not been previously demonstrated.
In contrast to the nutrient-rich BM milieu, the nutrient-poor LM are a harsh environment for survival. The adaptive mechanisms that allow tumors to thrive under cellular stressors within this niche are little understood. Furthermore, although the LM harbor a relative paucity of immune cells, they contain numerous resident macrophage populations. Macrophages are well-described to play protumoral roles in many tissues; yet, how BCCs might subvert meningeal macrophages to enhance their survival in the LM is only beginning to be investigated.
Metastases to the LM—the CSF-containing membranes surrounding the brain and spinal cord—occur in a wide variety of hematologic and solid malignancies, including leukemia, lymphoma, BC, lung cancer, and melanoma. When LM metastases arise, they are often rapidly fatal. The molecular mechanisms that enable LM metastasis are poorly understood, and there are limited interventions to prevent or treat this deadly disease complication.
By applying a combination of intravital and ex vivo three-dimensional confocal microscopy, micro–computed tomography (micro-CT), and histologic analyses to mouse models of bone-metastatic BC and LM metastasis, we demonstrated that BCCs could traffic to the LM from the BM through abluminal EV migration. We also found that BC cell surface expression of integrin α6, a laminin receptor, was essential for this process. Engraftment of mice with BC cells with CRISPR-mediated deletion of α6 inhibited LM colonization, decreased LMD development, and prolonged survival. Conversely, induced expression of α6 in BCCs increased LMD.
Imaging also demonstrated that the majority of BCCs colocalized with macrophages after entering the LM and that their presence stimulated macrophage secretion of the neuronal prosurvival molecule, glial-derived neurotrophic factor (GDNF). GDNF is minimally expressed in the healthy adult brain and LM but is secreted by reactive CNS microglia and macrophages in response to brain injury, where it is deposited in the extracellular matrix and serves to block apoptotic neuronal stress responses. Echoing this role in neurons, we found that BCCs that express the GDNF receptor, neural cell adhesion molecule (NCAM), can transduce antiapoptotic signaling that enhances their survival amid nutrient deprivation. Intrathecal GDNF blockade, macrophage-specific GDNF ablation, or deletion of NCAM from BCCs inhibited BC growth within the LM. Lastly, immunohistochemical analysis of patient samples showed that α6 expression was associated with meningeal-based metastases and that these metastases were highly enriched for BC-NCAM and stromal-GDNF expression.
Our data provide evidence that BCCs hijack a hematopoietic migration pathway to enter the LM via the BM. We also show that BCCs, mirroring neurons under stress, co-opt meningeal macrophages to aid their survival. Our findings describe previously unrecognized roles for GDNF and integrin α6 signaling in promoting BC LMD and thus provide the foundation for predictive, preventative, and therapeutic approaches to BC LM metastasis management.
The molecular mechanisms that regulate breast cancer cell (BCC) metastasis and proliferation within the leptomeninges (LM) are poorly understood, which limits the development of effective therapies. In this work, we show that BCCs in mice can invade the LM by abluminal migration along blood vessels that connect vertebral or calvarial bone marrow and meninges, bypassing the blood-brain barrier. This process is dependent on BCC engagement with vascular basement membrane laminin through expression of the neuronal pathfinding molecule integrin α6. Once in the LM, BCCs colocalize with perivascular meningeal macrophages and induce their expression of the prosurvival neurotrophin glial-derived neurotrophic factor (GDNF). Intrathecal GDNF blockade, macrophage-specific GDNF ablation, or deletion of the GDNF receptor neural cell adhesion molecule (NCAM) from BCCs inhibits breast cancer growth within the LM. These data suggest integrin α6 and the GDNF signaling axis as new therapeutic targets against breast cancer LM metastasis.
乳腺癌转移至软脑膜(LM)的治疗效果极差。软脑膜是含有脑脊液的膜性结构,覆盖于大脑和脊髓表面。Whiteley等人利用乳腺癌软脑膜转移的实验模型表明,表达细胞表面受体整合素α6的乳腺癌能够通过穿越血管外表面侵入软脑膜。这些血管通常将相邻的椎骨和颅骨骨髓与中枢神经系统脑膜相连接(参见Monteran和Erez的观点文章)。随后,乳腺癌细胞刺激驻留的脑膜巨噬细胞分泌一种称为胶质细胞源性神经营养因子的神经元促存活蛋白,该蛋白支持肿瘤生长。这些发现为软脑膜内的肿瘤-宿主相互作用提供了新见解,并可能指导针对软脑膜转移治疗的新靶点开发。—Priscilla N. Kelly
我们课题组先前发现,造血谱系细胞能够通过沿着穿静脉(EVs)富含层粘连蛋白的外表面迁移,从椎骨或颅骨骨髓(BM)移动到软脑膜(LM)。这些穿静脉是椎骨-颅骨骨髓脉管系统的一部分,它们穿过骨表面的孔,形成软脑膜血管,从而直接将骨髓腔室与中枢神经系统(CNS)连接起来。这种腔外血管迁移途径使得细胞无需进入血液循环、穿越中枢神经系统血脑屏障或血-脑脊液(CSF)屏障,也无需破坏组织边界。
骨髓是乳腺癌(BC)最常见的转移部位,并且大多数被诊断患有软脑膜疾病(LMD)的乳腺癌患者已形成椎骨转移。这一观察结果表明,穿静脉运输可能是乳腺癌细胞(BCCs)进入软脑膜的有效机制;然而,此前尚未有证据表明癌细胞能够通过穿静脉运输。
与营养丰富的骨髓微环境相比,营养贫乏的软脑膜是细胞生存的严酷环境。对于肿瘤如何在此生态位内的细胞应激条件下茁壮成长的适应性机制,人们知之甚少。此外,尽管软脑膜内的免疫细胞相对稀少,但它们含有大量的驻留巨噬细胞群体。众所周知,巨噬细胞在许多组织中发挥着促肿瘤作用;然而,关于乳腺癌细胞如何可能利用脑膜巨噬细胞以增强其在软脑膜中的存活,相关研究才刚刚开始。
转移至软脑膜——即包围大脑和脊髓的含脑脊液膜性结构——可发生于多种血液系统和实体恶性肿瘤,包括白血病、淋巴瘤、乳腺癌、肺癌和黑色素瘤。当发生软脑膜转移时,常常迅速致命。目前对驱动软脑膜转移的分子机制了解甚少,并且用于预防或治疗这种致命疾病并发症的干预手段有限。
通过结合活体内和离体三维共聚焦显微镜、微型计算机断层扫描(micro-CT)以及组织学分析,应用于骨转移性乳腺癌和软脑膜转移的小鼠模型,我们证明了乳腺癌细胞能够通过腔外穿静脉迁移的方式,从骨髓运输到软脑膜。我们还发现,乳腺癌细胞表面表达整合素α6(一种层粘连蛋白受体)对这一过程至关重要。通过CRISPR介导的α6基因敲除的乳腺癌细胞移植到小鼠体内,抑制了软脑膜定植,减少了软脑膜疾病的发展,并延长了生存期。相反,在乳腺癌细胞中诱导表达α6则增加了软脑膜疾病的发生。
成像分析还表明,大多数乳腺癌细胞在进入软脑膜后与巨噬细胞共定位,并且它们的存在刺激了巨噬细胞分泌神经元促存活分子——胶质细胞源性神经营养因子(GDNF)。GDNF在健康的成年大脑和软脑膜中表达极微,但由反应性中枢神经系统小胶质细胞和巨噬细胞响应脑损伤而分泌,随后沉积于细胞外基质中,起到阻断神经元凋亡应激反应的作用。与在神经元中的这种作用相呼应,我们发现,表达GDNF受体(神经细胞黏附分子,NCAM)的乳腺癌细胞能够转导抗凋亡信号,从而在营养匮乏的环境中增强其存活。鞘内阻断GDNF、巨噬细胞特异性敲除GDNF或从乳腺癌细胞中删除NCAM,均能抑制乳腺癌在软脑膜内的生长。最后,对患者样本的免疫组织化学分析表明,α6表达与基于脑膜的转移相关,并且这些转移灶中乳腺癌细胞的NCAM表达和基质中的GDNF表达高度富集。
我们的数据提供了证据,表明乳腺癌细胞劫持了一种造血迁移途径,通过骨髓进入软脑膜。我们还发现,乳腺癌细胞模仿处于应激状态下的神经元,利用脑膜巨噬细胞来辅助其生存。我们的研究结果揭示了GDNF和整合素α6信号在促进乳腺癌软脑膜疾病中先前未被认识的作用,从而为乳腺癌软脑膜转移管理的预测、预防和治疗方法的开发奠定了基础。
调控乳腺癌细胞(BCCs)在软脑膜(LM)内转移和增殖的分子机制尚不明确,这限制了有效疗法的开发。本研究中,我们表明小鼠体内的乳腺癌细胞能够通过沿着连接椎骨或颅骨骨髓与脑膜的血管进行腔外迁移来侵入软脑膜,从而绕过血脑屏障。这一过程依赖于乳腺癌细胞通过表达神经元路径寻找分子整合素α6,与血管基底膜层粘连蛋白的相互作用。一旦进入软脑膜,乳腺癌细胞便与血管周脑膜巨噬细胞共定位,并诱导其表达促存活的神经营养因子——胶质细胞源性神经营养因子(GDNF)。鞘内阻断GDNF、巨噬细胞特异性敲除GDNF或从乳腺癌细胞中删除GDNF受体神经细胞黏附分子(NCAM),均能抑制乳腺癌在软脑膜内的生长。这些数据提示,整合素α6和GDNF信号轴可作为对抗乳腺癌软脑膜转移的新治疗靶点。
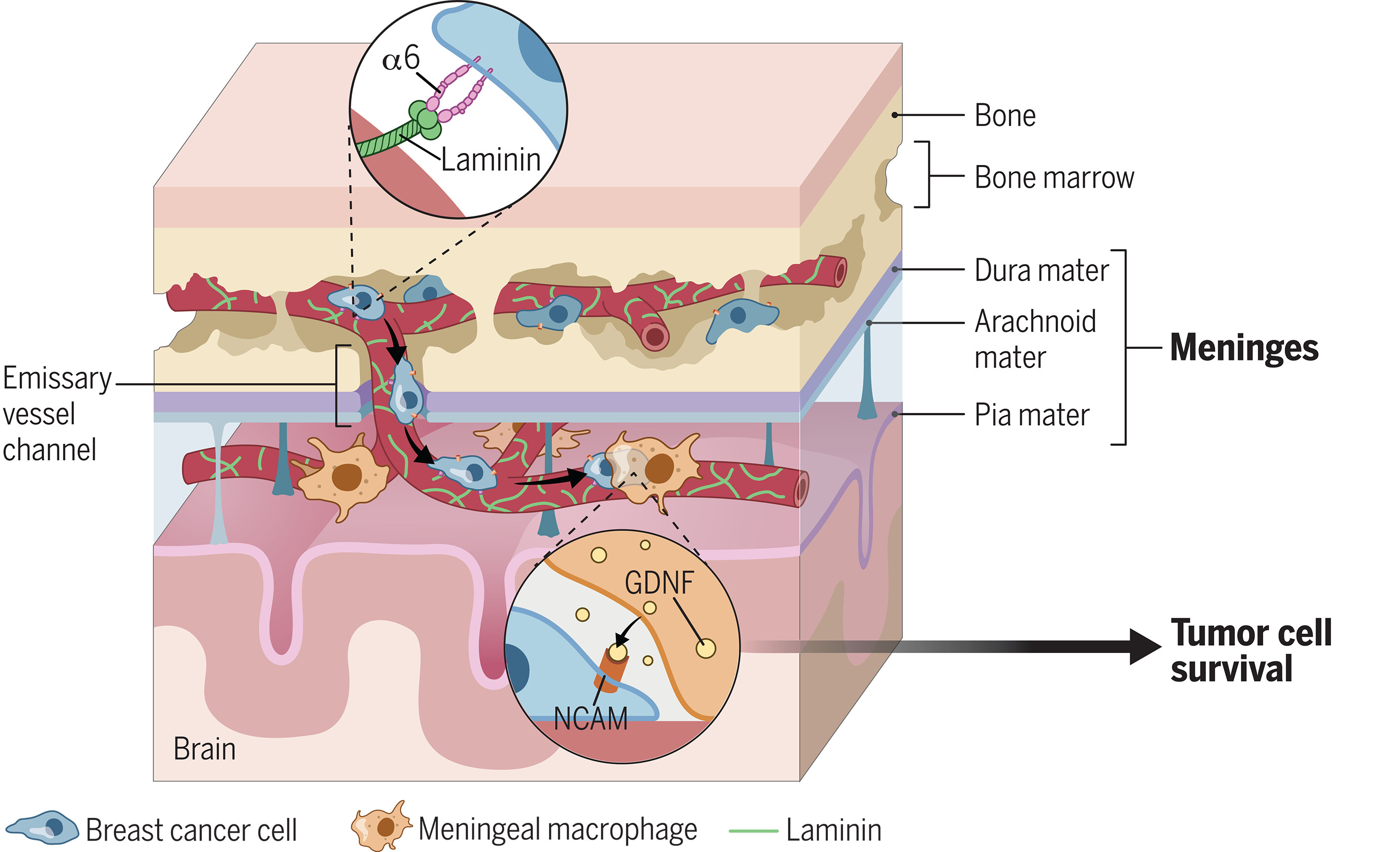
图:基底细胞癌在脑膜的转移及促生存相互作用。 表达整合素α6亚基(一种细胞表面受体,能使细胞黏附于基质分子层粘连蛋白)的基底细胞癌,可沿着富含层粘连蛋白的基底膜-软脑膜桥接穿通血管外表面迁移,进而侵入软脑膜。随后,基底细胞癌与脑膜巨噬细胞共定位,刺激其分泌胶质细胞源性神经营养因子;该因子与基底细胞癌的神经细胞黏附分子受体结合,进而激活生存信号通路。
Breast cancer exploits neural signaling pathways for bone-to-meninges metastasis