Inflammation of the pancreas, known as pancreatitis, can influence the emergence and progression of pancreatic cancer. How the body’s immune system responds to pancreatitis versus pancreatic cancer remains unclear. Mahadevan et al. report that pancreatitis leads to the recruitment of a class of immune cells called conventional dendritic cells that can help to protect tissue from self-destruction by T lymphocytes. They found that the coexistence of pancreatitis and pancreatic cancer resulted in an aggressive disease in which conventional dendritic cells influenced a suppressive T cell subset to override the action of T cells that usually destroy cancer. Blocking the suppressor T cells with immunotherapy reduced the growth of pancreatic cancer. Therefore, engineered cancer-specific conventional dendritic cells along with immunotherapy may provide a mechanism to intervene in pancreatic cancer. —Priscilla N. Kelly
Sites of chronic tissue injury and inflammation demonstrate an increased propensity to develop cancers. Tissue damage associated with chronic pancreatitis increases the risk of pancreatic ductal adenocarcinoma (PDAC). Chronic pancreatitis can precede the development of PDAC or occur concurrently during cancer progression due to bile or pancreatic duct involvement. However, the impact of pancreatitis on the immune microenvironment in PDAC remains unknown. In this study, we identify fundamental differences in the immune infiltrate of PDAC concurrent with and without pancreatitis. Pancreatitis enables recruitment of antigen-presenting dendritic cells and alters adaptive immunity associated with PDAC. Unexpectedly, this profile of immune microenvironment primes immunotherapy with elimination of pancreatic cancer.
Resistance to immune checkpoint blockade therapy (iCBT) has been attributed to a paucity of dendritic cells (DCs) and suppressive T cell response in PDAC. We evaluated whether parenchymal tissue injury and inflammation occurring in the background of cancer exhibit a different immune microenvironment and alter efficacy of iCBT.
Pancreatitis resulted in the recruitment of myeloid infiltrates into pancreas, accompanied by a suppression of T cells. Specifically, type 1 conventional dendritic cells (cDC1s) recruited in this setting presented self-antigens, which led to suppression of CD4+ and CD8+ T cell infiltrates to temper autoimmune tissue destruction. Mice depleted of cDC1s (XCR1 knockout mice) demonstrated elevated pancreatic acinar damage due to an increase in CD4+ Foxp3- effector T cells.
In PDAC associated with pancreatitis (ptPDAC), acceleration of tumor progression with an increase in activated cDC1s were observed. However, unlike in pancreatitis, CD4+ and CD8+ T cell suppression was not observed in ptPDAC. Activated CD11c+ DCs induced a tolerogenic CD4+ T cell response to accelerate tumor initiation in ptPDAC. Specific depletion of CD4+ T cells in ptPDAC resulted in robust CD8+ cytotoxic T cell responses and suppressed tumor initiation. Further, depletion of the CD11c+ DCs and CD4+ T cells together restored tumor initiation in ptPDAC, highlighting the importance of DCs in mediating CD8+ T cell responses in this context.
The cDC1s recruited in ptPDAC opened a therapeutic window and rendered tumors sensitive to iCBT. Ex vivo engineered cDC1s stimulated with tumor antigens sensitized PDAC to iCBT, with increased survival of PDAC-bearing mice. Further, the cDC1 vaccine with iCBT resulted in a functional CD8+ T cell memory response, and tumor rechallenge failed to induce PDAC.
We discovered an inherent difference in the immune microenvironment between PDAC and ptPDAC. Although ptPDAC accelerated disease progression, it is associated with a conducive immune profile that primes effective immunotherapy. Our data suggest that ptPDAC could benefit from iCBT and provides a proof of concept for the development of cDC1 vaccine for the treatment of PDAC in combination with iCBT.
Inflammation and tissue damage associated with pancreatitis can precede or occur concurrently with pancreatic ductal adenocarcinoma (PDAC). We demonstrate that in PDAC coupled with pancreatitis (ptPDAC), antigen-presenting type I conventional dendritic cells (cDC1s) are specifically activated. Immune checkpoint blockade therapy (iCBT) leads to cytotoxic CD8+ T cell activation and elimination of ptPDAC with restoration of life span even upon PDAC rechallenge. Using PDAC antigen-loaded cDC1s as a vaccine, immunotherapy-resistant PDAC was rendered sensitive to iCBT with elimination of tumors. cDC1 vaccination coupled with iCBT identified specific CDR3 sequences in the tumor-infiltrating CD8+ T cells with potential therapeutic importance. This study identifies a fundamental difference in the immune microenvironment in PDAC concurrent with, or without, pancreatitis and provides a rationale for combining cDC1 vaccination with iCBT as a potential treatment option.
胰腺的炎症,称为胰腺炎,可以影响胰腺癌的出现和进展。身体的免疫系统如何响应胰腺炎与胰腺癌仍不清楚。Mahadevan等人报告说,胰腺炎导致一类称为常规树突状细胞的免疫细胞的招募,这些细胞可以帮助保护组织免受T淋巴细胞的自毁。他们发现,胰腺炎和胰腺癌的共存导致了一种侵袭性疾病,其中常规树突状细胞影响了一个抑制性T细胞亚群,覆盖了通常破坏癌症的T细胞的作用。用免疫疗法阻断抑制性T细胞减少了胰腺癌的生长。因此,工程化的癌症特异性常规树突状细胞与免疫疗法结合可能提供干预胰腺癌的机制。—Priscilla N. Kelly
慢性组织损伤和炎症的部位显示出发展癌症的倾向增加。与慢性胰腺炎相关的组织损伤增加了胰腺导管腺癌(PDAC)的风险。慢性胰腺炎可以在PDAC发展之前发生,或在癌症进展期间由于胆管或胰管受累而同时发生。然而,胰腺炎对PDAC免疫微环境的影响仍然未知。在这项研究中,我们确定了PDAC伴随和不伴随胰腺炎的免疫浸润的基本差异。胰腺炎使得抗原呈递树突状细胞的招募成为可能,并改变了与PDAC相关的适应性免疫。出乎意料的是,这种免疫微环境的特征为免疫疗法消除了胰腺癌奠定了基础。
对免疫检查点阻断疗法(iCBT)的抵抗归因于PDAC中树突状细胞(DCs)的缺乏和抑制性T细胞反应。我们评估了在癌症背景下发生的实质组织损伤和炎症是否表现出不同的免疫微环境并改变iCBT的疗效。
胰腺炎导致骨髓浸润招募到胰腺,伴随着T细胞的抑制。具体来说,在这种背景下招募的1型常规树突状细胞(cDC1s)呈递自身抗原,这导致CD4+和CD8+ T细胞浸润的抑制,以缓和自身免疫组织破坏。cDC1s耗竭的小鼠(XCR1敲除小鼠)显示出胰腺腺泡损伤增加,由于CD4+ Foxp3-效应T细胞的增加。
在与胰腺炎相关的PDAC(ptPDAC)中,观察到肿瘤进展加速,伴随着活化的cDC1s增加。然而,与胰腺炎不同,在ptPDAC中未观察到CD4+和CD8+ T细胞的抑制。活化的CD11c+ DCs诱导了耐受性CD4+ T细胞反应,以加速ptPDAC中的肿瘤发生。在ptPDAC中特异性耗竭CD4+ T细胞导致强大的CD8+细胞毒性T细胞反应并抑制肿瘤发生。此外,同时耗竭CD11c+ DCs和CD4+ T细胞恢复了ptPDAC中的肿瘤发生,突出了DCs在这种背景下介导CD8+ T细胞反应的重要性。
在ptPDAC中招募的cDC1s打开了治疗窗口,并使肿瘤对iCBT敏感。体外工程化的cDC1s用肿瘤抗原刺激后,使PDAC对iCBT敏感,携带PDAC的小鼠生存期增加。此外,cDC1疫苗与iCBT结合导致功能性CD8+ T细胞记忆反应,肿瘤再次挑战未能诱导PDAC。
我们发现了PDAC和ptPDAC之间免疫微环境的内在差异。虽然ptPDAC加速了疾病进展,但它与一个有利于免疫疗法的免疫特征相关。我们的数据表明ptPDAC可能受益于iCBT,并为开发cDC1疫苗联合iCBT治疗PDAC提供了概念验证。
与胰腺炎相关的炎症和组织损伤可以在胰腺导管腺癌(PDAC)之前发生或同时发生。我们证明,在PDAC伴随胰腺炎(ptPDAC)的情况下,抗原呈递I型常规树突状细胞(cDC1s)被特异性激活。免疫检查点阻断疗法(iCBT)导致细胞毒性CD8+ T细胞激活并消除ptPDAC,即使在PDAC再次挑战时也能恢复寿命。使用PDAC抗原负载的cDC1s作为疫苗,免疫疗法抵抗的PDAC变得对iCBT敏感,并消除了肿瘤。cDC1疫苗接种与iCBT结合,在肿瘤浸润CD8+ T细胞中识别出具有潜在治疗重要性的特定CDR3序列。这项研究确定了PDAC伴随或不伴随胰腺炎的免疫微环境的基本差异,并为将cDC1疫苗接种与iCBT结合作为潜在治疗选择提供了理论依据。
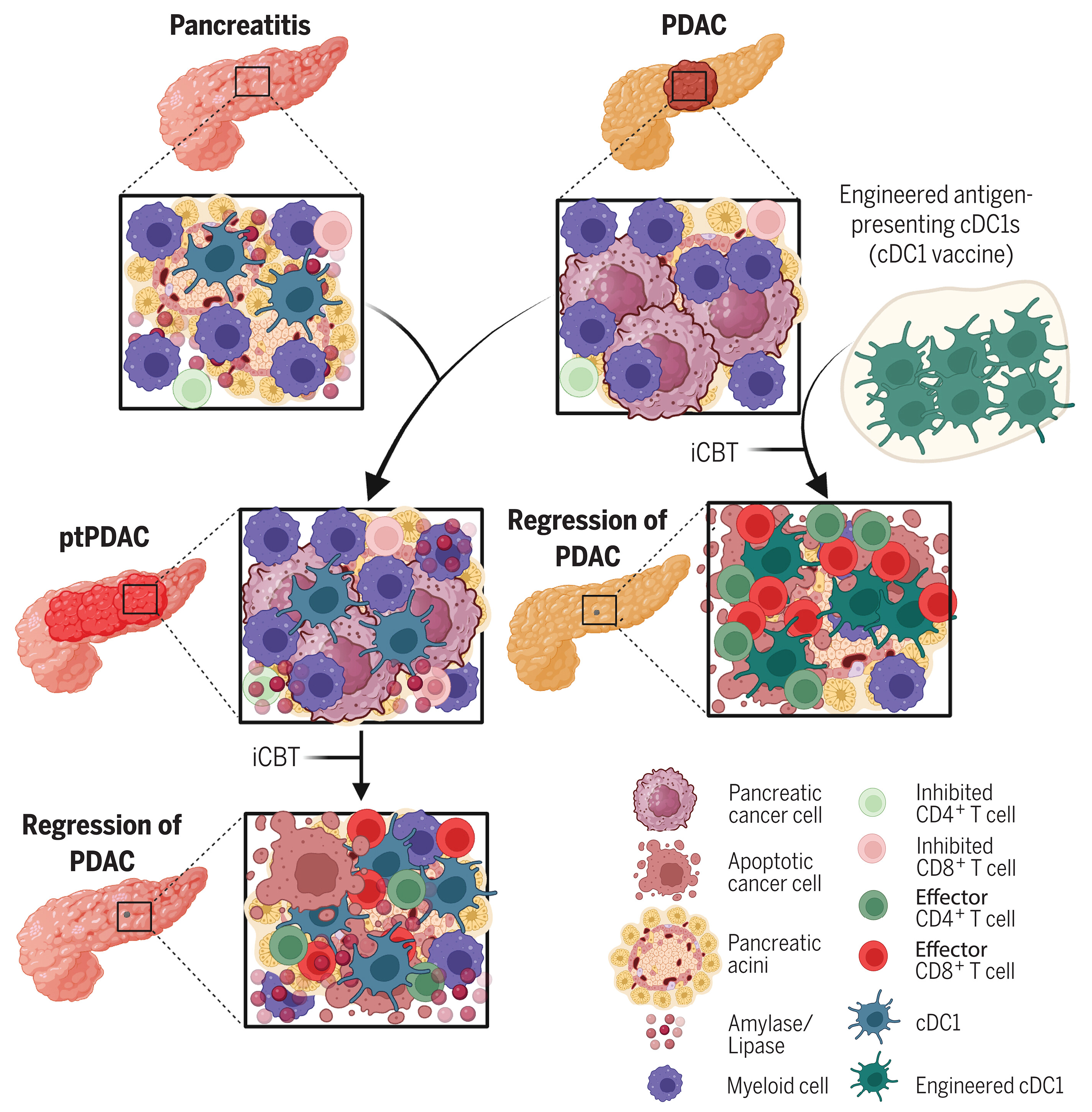
图:组织损伤与炎症影响cDC1疫苗对免疫治疗的启动作用。胰腺炎招募活化的cDC1进入胰腺,导致T细胞抑制(左上方)。PDAC的特征是髓系细胞浸润、抗原呈递性DC缺乏及效应T细胞稀少(右上方)。与PDAC相关的胰腺炎(左中)及抗原特异性cDC1疫苗给药(右中),联合iCBT治疗可引发效应性与记忆性T细胞反应,并消除PDAC(底部)。
Type I conventional dendritic cells facilitate immunotherapy in pancreatic cancer