Cancer immunotherapy is a type of treatment that mobilizes a patient’s immune system to kill tumor cells. It has been successful in treating certain tumors, but patients frequently have chronic inflammation and immunosuppression, which can limit treatment response. Two independent clinical trials looked at whether dialing down inflammation using drugs called JAK inhibitors could improve the efficacy of anti–PD-1 immunotherapy in cancer patients (see the Perspective by Gadina and O’Shea). Mathew et al. conducted a phase 2 trial to investigate the drug combination as a first-line therapy for metastatic non-small-cell lung cancer. Delayed administration of itacitinib after treatment with pembrolizumab improved therapeutic response. Zak et al. performed a phase 1/2 trial in patients with relapsed/refractory Hodgkin’s lymphoma. A combination of ruxolitinib and nivolumab resulted in improved clinical efficacy in patients who had previously failed checkpoint blockade immunotherapy. —Priscilla N. Kelly
Inflammation is a hallmark of cancer but is also required to generate optimal antitumor immune responses. Although short exposure to cytokines such as interferon (IFN) can enhance tumor immunity, prolonged exposure can result in immunosuppression. The dual nature of inflammation makes it challenging to harness the beneficial effects of cytokine activation during cancer immunotherapy while avoiding detrimental consequences. Thus, a therapeutical strategy to effectively modulate these often-opposing functions of cytokine signaling could improve immunotherapy efficacy and mitigate development of resistance.
Preclinical studies in mice have demonstrated that blockade of type-one IFN (IFN-I) signaling can improve immune function during chronic viral infections and enhance the efficacy of immunotherapy for cancer. Moreover, tumors from patients with lung and other cancer types that relapse after—or are resistant to—immune checkpoint blockade (ICB) can have high expression of IFN-stimulated genes. Thus, we conducted a phase 2 clinical trial for first-line metastatic non–small cell lung cancer (NSCLC) with programmed death ligand 1 (PD-L1) expression ≥50%. Patients were administered 6 weeks of anti–PD-1 (PD-1, programmed cell death protein 1) immunotherapy, followed by a combination of anti–PD-1 and itacitinib [a selective Janus kinase 1 (JAK1) inhibitor] for 6 weeks before continuing with anti–PD-1 alone. To understand the immunological effects of transient JAK inhibition aimed at interfering with persistent inflammation occurring after anti–PD-1, we evaluated the association between clinical response and the evolution of CD8 T cell differentiation, immune signaling, and inflammatory markers.
In mice, the addition of itacitinib after the start of ICB improved response of interferon-stimulated gene (ISG)–high resistant tumors and increased the proportion of proliferating precursor-like CD8 T cells in the periphery. Use of an IFN-I–receptor blocking antibody similarly improved ICB efficacy, suggesting that blockade of IFN-I signaling is sufficient to phenocopy the effects of delayed JAK inhibition. In humans, the combination of anti–PD-1 with delayed, transient, itactinib treatment in patients with NSCLC led to an overall response rate of 67% and median progression-free survival of 23.8 months. Patients were categorized into three response groups based on the timing of radiographic response: patients with clinical response within the first two cycles of anti–PD-1 monotherapy (αPD1.R), patients who responded only after a 6-week course of concurrent itacitinib was added at the start of the third cycle of anti–PD-1 (JAKi.R), or patients who did not respond regardless of treatment (NR). Each clinical response group had distinctive immunological changes coupled to inflammatory features. The αPD1.R patients had low baseline inflammation and CD8 T cell responses after anti–PD-1 alone. JAKi.R patients had elevated inflammatory markers, poor CD8 T cell responses, and blunted immune signaling after anti–PD-1 alone. However, after addition of itacitinib, subsequent clinical responses in JAKi.R patients were associated with decreased inflammatory signaling accompanied by an increase in a “fate-flexible” CD8 T cell progenitor-like population. This fate-flexible population was linked to features of greater CD8 T cell plasticity. By contrast, NR patients had high baseline inflammation refractory to JAK inhibition. This persistent inflammatory and IFN-I signaling in NR patients was associated with CD8 T cell terminal differentiation and treatment failure.
As a therapeutic strategy to block the immunosuppressive effects of persistent IFN and/or chronic inflammation, JAK inhibition after initial anti–PD-1 is safe, feasible, and associated with durable and high response rates in NSCLC. JAK inhibition may be particularly beneficial in patients with elevated inflammation who have poor CD8 T cell responses to anti–PD-1 alone. In this study, JAK inhibition enhanced CD8 T cell plasticity by decreasing the inflammatory and IFN-I signals that drive terminal differentiation of CD8 T cells. However, some patients with the highest baseline levels of inflammation were largely unaffected by JAK1 inhibition and had progressive terminal CD8 T cell differentiation and disease progression. Our findings suggest that JAK inhibition can target chronic immunoregulatory functions of cytokine signaling that contribute to relapse during cancer immunotherapy and is a strategy warranting further preclinical and clinical investigation.
Persistent inflammation driven by cytokines such as type-one interferon (IFN-I) can cause immunosuppression. We show that administration of the Janus kinase 1 (JAK1) inhibitor itacitinib after anti–PD-1 (programmed cell death protein 1) immunotherapy improves immune function and antitumor responses in mice and results in high response rates (67%) in a phase 2 clinical trial for metastatic non–small cell lung cancer. Patients who failed to respond to initial anti–PD-1 immunotherapy but responded after addition of itacitinib had multiple features of poor immune function to anti–PD-1 alone that improved after JAK inhibition. Itacitinib promoted CD8 T cell plasticity and therapeutic responses of exhausted and effector memory–like T cell clonotypes. Patients with persistent inflammation refractory to itacitinib showed progressive CD8 T cell terminal differentiation and progressive disease. Thus, JAK inhibition may improve the efficacy of anti–PD-1 immunotherapy by pivoting T cell differentiation dynamics.
癌症免疫疗法是一种调动患者免疫系统以杀死肿瘤细胞的治疗方法。它在治疗某些肿瘤方面取得了成功,但患者常常存在慢性炎症和免疫抑制,这可能会限制治疗反应。两项独立的临床试验研究了使用名为JAK抑制剂的药物来减轻炎症是否能提高癌症患者抗PD-1免疫疗法的疗效(参见Gadina和O'Shea的观点文章)。Mathew等人进行了一项2期试验,研究该药物组合作为转移性非小细胞肺癌的一线疗法。在帕博利珠单抗治疗后延迟给予伊他替尼改善了治疗反应。Zak等人在复发/难治性霍奇金淋巴瘤患者中进行了一项1/2期试验。鲁索替尼和纳武利尤单抗的联合治疗在先前检查点阻断免疫疗法失败的患者中改善了临床疗效。—Priscilla N. Kelly
炎症是癌症的一个标志,但同时也是产生最佳抗肿瘤免疫反应所必需的。虽然短期暴露于干扰素(IFN)等细胞因子可以增强肿瘤免疫,但长期暴露可能导致免疫抑制。炎症的这种双重性质使得在癌症免疫治疗中,既要利用细胞因子激活的有益效果,又要避免其有害后果,变得具有挑战性。因此,一种能有效调节细胞因子信号传导这些常常相反功能的治疗策略,可能会提高免疫疗法的疗效并减轻耐药性的发展。
小鼠临床前研究表明,阻断I型干扰素(IFN-I)信号传导可以在慢性病毒感染期间改善免疫功能,并提高癌症免疫疗法的疗效。此外,对免疫检查点阻断(ICB)后复发或耐药的肺癌和其他癌症类型患者的肿瘤,可能具有高表达的IFN刺激基因。因此,我们针对程序性死亡配体1(PD-L1)表达≥50%的一线转移性非小细胞肺癌(NSCLC)进行了一项2期临床试验。患者接受6周的抗PD-1(PD-1,程序性细胞死亡蛋白1)免疫治疗,随后在继续单独使用抗PD-1之前,进行6周的抗PD-1联合伊他替尼[一种选择性Janus激酶1(JAK1)抑制剂]治疗。为了理解旨在干扰抗PD-1治疗后发生的持续性炎症的短暂JAK抑制所产生的免疫学效应,我们评估了临床反应与CD8 T细胞分化、免疫信号传导和炎症标志物演变之间的关联。
在小鼠中,在ICB开始后添加伊他替尼改善了干扰素刺激基因(ISG)高表达耐药肿瘤的反应,并增加了外周血中增殖性前体样CD8 T细胞的比例。使用IFN-I受体阻断抗体同样提高了ICB疗效,这表明阻断IFN-I信号传导足以模拟延迟JAK抑制的效果。在人类中,对于NSCLC患者,抗PD-1联合延迟、短暂的伊他替尼治疗导致总缓解率达到67%,中位无进展生存期为23.8个月。根据影像学反应出现的时间,将患者分为三个反应组:在最初两个周期的抗PD-1单药治疗期间出现临床反应的患者(αPD1.R)、仅在抗PD-1第三周期开始时加用为期6周的伊他替尼联合治疗后才有反应的患者(JAKi.R),或无论何种治疗均无反应的患者(NR)。每个临床反应组都具有与炎症特征相关的独特免疫学变化。αPD1.R患者基线炎症水平低,并且在单独抗PD-1治疗后出现CD8 T细胞反应。JAKi.R患者在单独抗PD-1治疗后炎症标志物升高、CD8 T细胞反应差且免疫信号传导减弱。然而,在加用伊他替尼后,JAKi.R患者随后的临床反应与炎症信号传导减少相关,同时伴随“命运可塑的”CD8 T细胞祖细胞样群体的增加。这种命运可塑的群体与更强的CD8 T细胞可塑性特征相关。相比之下,NR患者具有对JAK抑制无效的高基线炎症水平。NR患者中这种持续的炎症和IFN-I信号传导与CD8 T细胞终末分化和治疗失败相关。
作为阻断持续性IFN和/或慢性炎症的免疫抑制效应的治疗策略,在初始抗PD-1治疗后进行JAK抑制在NSCLC中是安全、可行的,并且与持久和高缓解率相关。JAK抑制可能对炎症水平升高且对单独抗PD-1治疗CD8 T细胞反应不佳的患者特别有益。在这项研究中,JAK抑制通过减少驱动CD8 T细胞终末分化的炎症和IFN-I信号,增强了CD8 T细胞的可塑性。然而,一些基线炎症水平最高的患者,其病情在很大程度上不受JAK1抑制的影响,并出现了进展性的CD8 T细胞终末分化和疾病进展。我们的研究结果表明,JAK抑制可以靶向细胞因子信号传导中导致癌症免疫治疗期间复发的慢性免疫调节功能,是一种值得进一步临床前和临床研究的策略。
I型干扰素(IFN-I)等细胞因子驱动的持续性炎症可导致免疫抑制。我们发现,在抗PD-1(程序性细胞死亡蛋白1)免疫治疗后给予Janus激酶1(JAK1)抑制剂伊他替尼,可改善小鼠的免疫功能和抗肿瘤反应,并在针对转移性非小细胞肺癌的2期临床试验中获得了高缓解率(67%)。对初始抗PD-1免疫治疗无反应但在加用伊他替尼后有反应的患者,表现出多种对单独抗PD-1免疫功能不良的特征,这些特征在JAK抑制后得到改善。伊他替尼促进了CD8 T细胞的可塑性以及耗竭性和效应记忆样T细胞克隆型的治疗反应。对伊他替尼无效的持续性炎症患者表现出进行性的CD8 T细胞终末分化和疾病进展。因此,JAK抑制可能通过改变T细胞分化动态来提高抗PD-1免疫疗法的疗效。
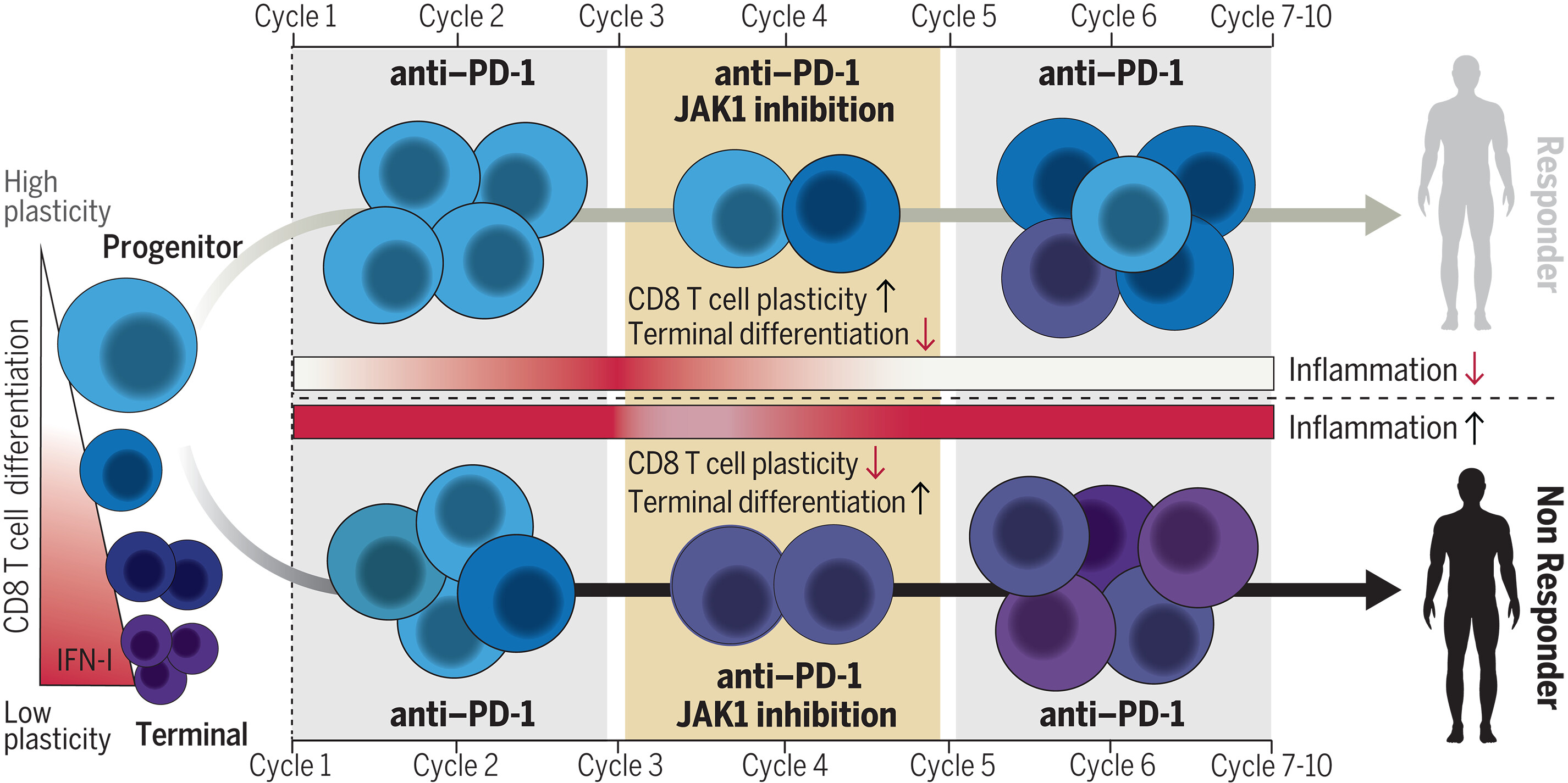
图:AQP5是小鼠和人类胃癌干细胞的功能标记物。我们建立了多种体外和体内胃癌模型。这些模型产生的肿瘤具有异质性,并包含AQP5阳性细胞亚群,该亚群可被选择性分离。多项实验表明AQP5阳性群体具有癌症干细胞潜能。此外,通过构建AQP5基因敲除和过表达细胞,我们揭示了AQP5在癌细胞增殖、侵袭和远处转移中的功能性作用。FACS,即荧光激活细胞分选技术。
Combined JAK inhibition and PD-1 immunotherapy for non–small cell lung cancer patients