γδ T cells are a unique population of immune cells that can recognize and kill tumors. They comprise an evolutionarily conserved T cell lineage that holds promise for next-generation immunotherapies aimed at reaching more diverse patient groups, possibly with fewer adverse events. Combining innate-like traits such as enrichment within tissues and rapid responsiveness, γδ T cells have broader reactivities than the αβ T cell subset. Furthermore, because γδ cells are not restricted by an individual’s major histocompatibility complex molecules, “off-the-shelf” biobanks of well-characterized therapeutic γδ cells have the capacity to be generated from healthy donors. Hayday et al. discuss current research efforts focused on how γδ cells naturally discriminate cancers from healthy tissues, explore the advantages of γδ T cell immunotherapy, and consider the progress and prospects of several γδ T cell–based clinical trials and applications. —Priscilla N. Kelly
This century has seen game-changing progress in the delivery of αβ T cell–centric cancer immunotherapies, including immune checkpoint blockade, chimeric antigen receptor (CAR) T cells, tumor-infiltrating lymphocytes, and affinity-enhanced T cell receptors (TCRs) in soluble and membrane-bound forms. Empowered by years of research elucidating key aspects of αβ T cell biology, clinicians have achieved notable rates of remission in patients with hitherto deadly diseases such as metastatic melanoma and multiple myeloma. However, there is much more to do. In particular, successful immunotherapy needs to reach increased numbers of patients of diverse ethnicities with a much broader range of tumor types. It needs to do so while incurring fewer adverse events and with the goal being complete cure.
An emerging approach to achieving this ambition is to exploit γδ T cells, which first came to light about 40 years ago and comprise a distinct lymphocyte lineage that is mostly conserved across jawed vertebrates. Fundamental research has highlighted many aspects of γδ T cell biology that seem very well suited to cancer immunotherapy, including the cells’ tropism for tissues in which solid tumors form, their very broad recognition potentials, their high tolerance of normal tissues, and their functional pleiotropy. In particular, γδ T cells seem able to combine highly specific adaptive responses with the rapidity and polyclonality of innate immunity, including a capacity to harness a battery of so-called natural killer receptors that target transformed cells. In this review, we consider these signature properties, their ongoing clinical application, and the emerging efficacy and safety readouts.
The γδ TCR is not restricted to the recognition of peptides presented by major histocompatibility complex (MHC) molecules. Hence, γδ T cells are not limited to perceiving so-called neoantigenic peptides that reflect high levels of genomic instability but should instead be able to target a wide range of tumors with low mutagenic loads across ethnically diverse patient populations not limited by MHC haplotype. That targeting is based on the capacity of TCRγδ to function like an antibody in recognizing diverse cell-surface antigens, many of which are monomorphic and overexpressed or misexpressed by a spectrum of different cancer cells. In the CAR-T cell format, MHC-independent recognition has already opened the door to trials that have delivered highly reproducible, off-the-shelf allogeneic γδ T cells that obviate the need for autologous grafts from patients whose immune systems are commonly greatly compromised. From more than 20 ongoing clinical trials, safety profiles, albeit at an early stage, have been promising, possibly in part reflecting a signature molecular recognition device, termed normality sensing, by which γδ T cells can discriminate cancer cells from healthy tissue. Additionally, the pleiotropic functions of distinct γδ T cell subsets include three that may be of particular importance in striving for cure, namely, a potent capacity to lyse tumor cells, the capacity to enhance CD8 αβ T cell memory, and the capacity to promote wound resolution.
Early reports of efficacy and safety from ongoing clinical applications of γδ T cells in different settings can now be built upon, extending their application to cancers ill-suited to αβ T cell–centric approaches, increasing the numbers of those who may benefit, and reducing costs by further refinement of off-the-shelf adoptive cell therapies and well-formulated cell engagers and regulators. Many lessons from αβ T cell immunotherapy may prove invaluable, but approaches going forward should nonetheless focus on preserving biological properties specific to γδ T cells. This in turn demands the support of more basic research in several areas, including ligand identification, better preclinical models for predicting the efficacy of various therapeutic modalities, improved cell expansion methods, and the identification of mechanisms of γδ T cell immunoevasion. Moreover, given rapidly developing cancer vaccine strategies, research should be directed toward better understanding and harnessing the apparent capacity of γδ T cells to promote vaccine immunogenicity.
The premise of cancer immunotherapy is that cancers are specifically visible to an immune system tolerized to healthy self. The promise of cancer immunotherapy is that immune effector mechanisms and immunological memory can jointly eradicate cancers and inoperable metastases and de facto vaccinate against recurrence. For some patients with hitherto incurable diseases, including metastatic melanoma, this promise is being realized by game-changing immunotherapies based on αβ T cells. Today’s challenges are to bring benefit to greater numbers of patients of diverse ethnicities, target more cancer types, and achieve a cure while incurring fewer adverse events. In meeting those challenges, specific benefits may be offered by γδ T cells, which compose a second T cell lineage with distinct recognition capabilities and functional traits that bridge innate and adaptive immunity. γδ T cell–based clinical trials, including off-the-shelf adoptive cell therapy and agonist antibodies, are yielding promising results, although identifiable problems remain. In addressing those problems, we advocate that immunotherapies be guided by the distinctive biology of γδ T cells, as elucidated by ongoing research.
γδ T细胞是一类独特的免疫细胞群,能够识别并杀伤肿瘤。它们构成了一个进化上保守的T细胞谱系,有望用于下一代免疫疗法,旨在惠及更广泛的患者群体,并可能减少不良事件。γδ T细胞兼具组织内富集和快速反应等先天样特性,其反应范围比αβ T细胞亚群更广。此外,由于γδ细胞不受个体主要组织相容性复合体分子的限制,可以从健康供体中建立特征明确、可用于治疗的γδ细胞“现货”生物库。Hayday等人讨论了当前聚焦于γδ细胞如何天然区分癌症与健康组织的研究工作,探讨了γδ T细胞免疫疗法的优势,并审视了几项基于γδ T细胞的临床试验及应用的进展与前景。——Priscilla N. Kelly
本世纪见证了以αβ T细胞为核心的癌症免疫疗法取得颠覆性进展,包括免疫检查点阻断、嵌合抗原受体T细胞、肿瘤浸润淋巴细胞以及可溶性和膜结合形式的亲和力增强型T细胞受体。得益于多年来阐明αβ T细胞生物学关键方面的研究,临床医生已在转移性黑色素瘤和多发性骨髓瘤等既往致命的疾病患者中实现了显著的缓解率。然而,仍有更多工作要做。特别是,成功的免疫疗法需要惠及更多不同族裔的患者,覆盖更广泛的肿瘤类型,同时减少不良事件的发生,并以完全治愈为目标。
实现这一目标的一个新兴方法是利用γδ T细胞。γδ T细胞大约在40年前被发现,构成了一个独特的淋巴细胞谱系,在有颌脊椎动物中大多保守存在。基础研究揭示了γδ T细胞生物学的许多方面似乎非常适合癌症免疫疗法,包括这些细胞对实体瘤形成组织的趋向性、其广泛的识别潜力、对正常组织的高耐受性以及其功能多效性。特别是,γδ T细胞似乎能够将高度特异性的适应性应答与先天免疫的快速性和多克隆性结合起来,包括利用一系列靶向转化细胞的所谓自然杀伤受体。在本综述中,我们探讨了这些特征性属性、其当前的临床应用以及新出现的疗效和安全性结果。
γδ TCR不受限于识别由主要组织相容性复合体分子呈递的肽段。因此,γδ T细胞不仅限于感知反映高水平基因组不稳定的所谓新抗原肽,而应能靶向多种突变负荷低的肿瘤,且不受MHC单倍型限制,可惠及不同族裔的患者群体。这种靶向作用基于TCRγδ能够像抗体一样识别多种细胞表面抗原的能力,其中许多抗原是单态的,并在多种不同癌细胞中过度表达或异常表达。在CAR-T细胞形式中,这种不依赖MHC的识别方式已经为临床试验打开了大门,这些试验提供了高度可重复的、现成的同种异体γδ T细胞,从而避免了通常免疫系统已严重受损的患者需要进行自体移植。从超过20项正在进行的临床试验来看,安全性数据(尽管处于早期阶段)一直令人鼓舞,这可能部分归因于一种被称为“正常性感知”的特征性分子识别机制,γδ T细胞借此能够区分癌细胞与健康组织。此外,不同γδ T细胞亚群的多效性功能包括三点可能对争取治愈尤为重要,即强大的溶解肿瘤细胞能力、增强CD8 αβ T细胞记忆的能力以及促进伤口愈合的能力。
目前,可以在γδ T细胞在不同情境下临床应用已取得的早期疗效和安全性报告基础上,将其应用扩展到不适合以αβ T细胞为核心方法的癌症,增加可能受益的患者数量,并通过进一步改进现成的过继性细胞疗法以及精心设计的细胞衔接器和调节剂来降低成本。从αβ T细胞免疫疗法中汲取的许多经验可能证明是无价的,但未来的方法仍应侧重于保留γδ T细胞特有的生物学特性。这反过来需要支持更多基础研究,包括配体鉴定、更好的临床前模型以预测各种治疗模式的疗效、改进细胞扩增方法以及识别γδ T细胞免疫逃逸机制。此外,鉴于癌症疫苗策略的快速发展,研究应致力于更好地理解和利用γδ T细胞在促进疫苗免疫原性方面的明显能力。
癌症免疫疗法的前提是,对健康自身耐受的免疫系统能够特异性识别癌症。癌症免疫疗法的承诺在于,免疫效应机制和免疫记忆可以联合根除癌症和无法手术的转移灶,并实际上起到预防复发的疫苗作用。对于一些既往无法治愈的疾病(包括转移性黑色素瘤)患者,这一承诺正在通过基于αβ T细胞的颠覆性免疫疗法实现。当前的挑战是让更多不同族裔的患者获益,靶向更多癌症类型,并在减少不良事件的同时实现治愈。为了应对这些挑战,γδ T细胞可能提供特定的益处。γδ T细胞构成了第二个T细胞谱系,具有独特的识别能力和连接先天免疫与适应性免疫的功能特性。基于γδ T细胞的临床试验,包括现成的过继性细胞疗法和激动性抗体,正在产生有希望的结果,尽管仍存在一些可识别的问题。为解决这些问题,我们主张免疫疗法应以正在进行的研究所阐明的γδ T细胞的独特生物学特性为指导。
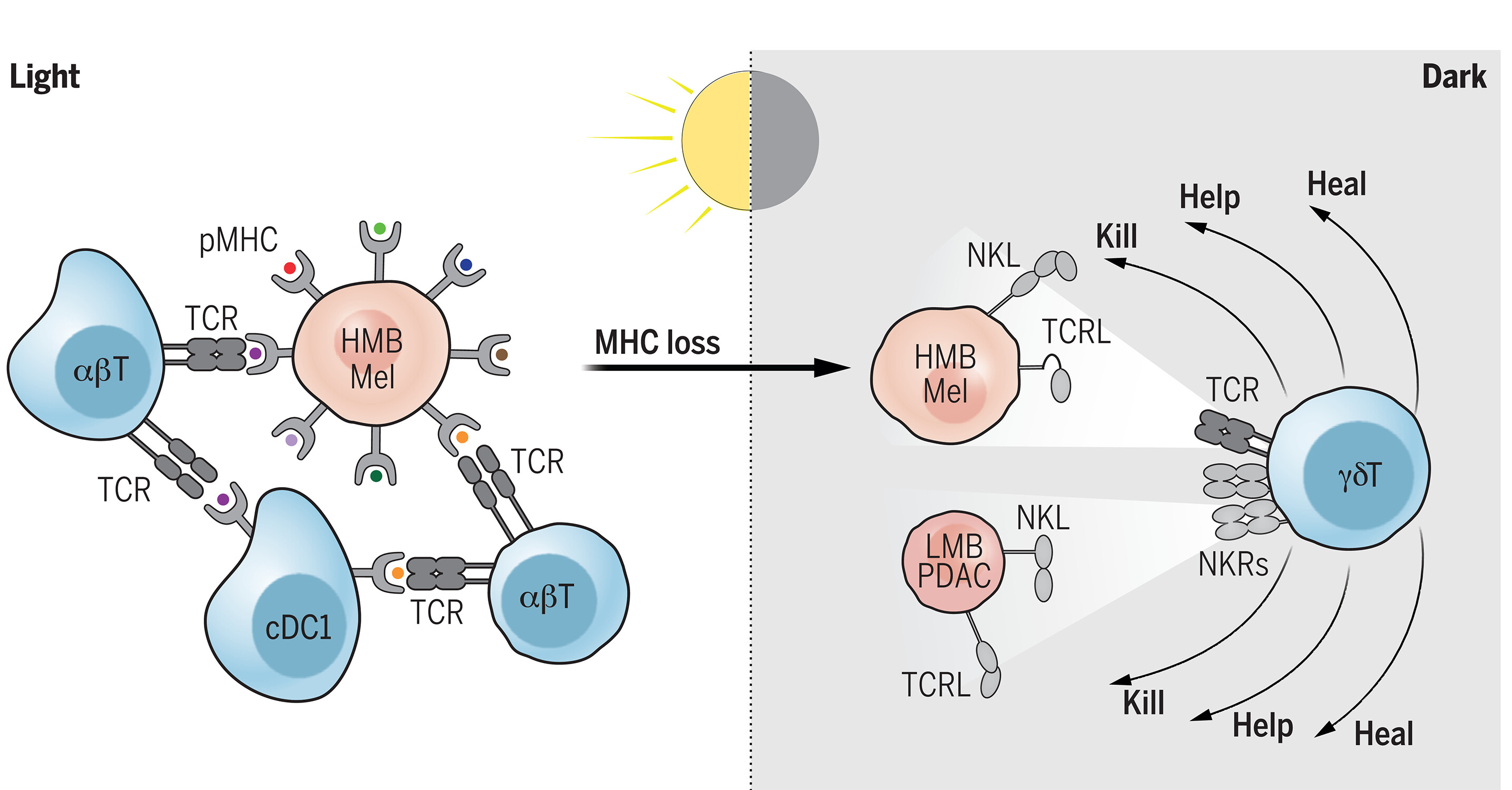
图:γδ T细胞照亮癌症免疫治疗的“阴暗面”。由αβ T细胞介导、针对MHC分子(杯状-柄状结构)呈递的新抗原肽的免疫监视已被证明非常成功,尤其对于黑色素瘤(Mel)等高突变负荷(HMB)的癌细胞。这一策略得益于对常规1型树突状细胞(cDC1)抗原呈递等免疫机制的深入理解。然而,癌细胞可通过下调MHC分子转入“阴暗面”,从而难以被αβ T细胞识别,低突变负荷(LMB)的癌细胞(如胰腺腺癌PDAC细胞)同样如此。但这类细胞可能被γδ T细胞检测到——γδ T细胞通过自然杀伤受体(NKRs)识别激活性的NKR配体(NKLs)和/或通过γδ TCR识别多样化的非MHC限制性TCR配体(TCRLs)。活化的γδ T细胞具有多效性潜能:可杀伤肿瘤靶细胞,辅助其他抗癌免疫反应,并促进伤口愈合,这些功能均可能助力癌症治愈。αβT指αβ T细胞;γδT指γδ T细胞。