Immune checkpoint blockade therapy for the treatment of cancer can have reduced efficacy when antibiotics are administered and then discontinued before the start of therapy. Fidelle et al. investigated whether bacteria that rebound may affect the treatment response (see the Perspective by Pratt and Milner). Enterocloster species that recolonized the guts of mice treated with antibiotics down-regulated the expression of MAdCAM-1, the ligand for integrin α4β7 that helps to retain an immunosuppressive subset of T cells (Tr17 cells) within the gut. This leads to transit of Tr17 cells to tumors and tumor-draining lymph nodes, where they compromise immune checkpoint blockade therapy. In cancer patients undergoing immunotherapy, low levels of serum-soluble MAdCAM-1 correlated with intestinal dysbiosis and poor clinical outcomes for renal, bladder, and lung tumors. —Priscilla N. Kelly & Seth Thomas Scanlon
Resistance of cancers to immune checkpoint inhibitors (ICIs) can result from antibiotic (ABX) treatment, likely as a result of a deviated gut microbiota. ABX compromise clinical outcome when administered before, rather than during, ICI administration, suggesting that bacterial recolonization following ABX discontinuation may be deleterious. Gut commensals induce the differentiation of an immunosuppressive subset of FoxP3+ retinoic acid receptor–related orphan receptor-γt (RORγt+) regulatory (Treg17) cells. Lymphocytes primed in the mesenteric lymph nodes (mLNs) or homing to the intestinal lamina propria express the α4β7 integrin interacting with its counter-receptor, mucosal addressin cell adhesion molecule-1 (MAdCAM-1), which is expressed in high endothelial venules (HEVs).
We hypothesized that disruption of the MAdCAM-1–α4β7 interaction that retains Treg17 cells might cause their migration from the gut to tumors and thereby compromise the anticancer effects of ICIs. We used two complementary methods to visualize the exodus of intestinal T cells to subcutaneous tumors and tumor-draining lymph nodes (tdLNs): (i) Kaede mice expressing a fluorescent protein that is photoconverted upon ultraviolet light illumination of the ileum and (ii) the injection of carboxyfluorescein succinimidyl ester into mLNs. Moreover, we used transgene-enforced Madcam1 expression in the liver to locally intercept Treg17 cells during their migration.
Several classes of ABX down-regulated Madcam1 expression in ileal venules, Peyer’s patches and mLNs, coinciding with the ileal exodus of α4β7+ T helper (Th17) and Treg17 cells toward extraintestinal tumors and tdLNs. This ABX-induced reduction in MAdCAM-1 could be explained by the recolonization of the gut by the genus Enterocloster (encompassing the E. clostridioformis species), because its oral administration was sufficient to down-regulate MAdCAM-1 expression through its effects on bile acid metabolism. Genetic or antibody-mediated neutralization of MAdCAM-1 or α4β7 integrin phenocopied the immunosuppressive effects of ABX, promoting resistance to ICIs targeting programmed cell death protein 1 (PD-1) and inducing a surge in gut-derived α4β7+ Treg17 cells in tdLNs and tumors. Restoration of MAdCAM-1 on ileal HEV by fecal microbial transplantation or blockade of IL-17A reversed the inhibitory effects of ABX. Ectopic expression of MAdCAM-1 in the liver caused the local retention of enterotropic α4β7+ Treg17 cells, reducing their accumulation in tumor beds and improving immunotherapy outcomes in mice. Finally, low-serum-soluble MAdCAM-1 was identified as a proxy of intestinal dysbiosis and a robust predictor of shorter overall and progression-free survival of renal, bladder, and lung cancer patients under immunotherapy with antibodies targeting PD-1 or PD-L1. In non-small-cell lung cancer patients, the prognostic value of soluble MAdCAM-1 was independent of PD-L1 expression.
The relocation of enterotropic and immunosuppressive Treg17 cells to cancerous tissue (tumors and tdLNs) is repressed by the molecular interaction between the HEV addressin MAdCAM-1 and the integrin α4β7 expressed by Treg17 cells. Disruption of the MAdCAM-1 expression by ABX or gut dysbiosis causes the relocation of Treg17 cells into tumors, consequently compromising cancer immunosurveillance and the therapeutic efficiency of ICIs in mice and patients.
Antibiotics (ABX) compromise the efficacy of programmed cell death protein 1 (PD-1) blockade in cancer patients, but the mechanisms underlying their immunosuppressive effects remain unknown. By inducing the down-regulation of mucosal addressin cell adhesion molecule 1 (MAdCAM-1) in the ileum, post-ABX gut recolonization by Enterocloster species drove the emigration of enterotropic α4β7+CD4+ regulatory T 17 cells into the tumor. These deleterious ABX effects were mimicked by oral gavage of Enterocloster species, by genetic deficiency, or by antibody-mediated neutralization of MAdCAM-1 and its receptor, α4β7 integrin. By contrast, fecal microbiota transplantation or interleukin-17A neutralization prevented ABX-induced immunosuppression. In independent lung, kidney, and bladder cancer patient cohorts, low serum levels of soluble MAdCAM-1 had a negative prognostic impact. Thus, the MAdCAM-1–α4β7 axis constitutes an actionable gut immune checkpoint in cancer immunosurveillance.
在癌症治疗中,若在免疫检查点阻断疗法开始前使用并随后停用抗生素,可能会降低该疗法的疗效。Fidelle等人研究了反弹的细菌是否可能影响治疗反应(参见Pratt和Milner的观点)。在用抗生素治疗的小鼠肠道中重新定植的肠梭菌属物种下调了MAdCAM-1的表达,MAdCAM-1是整合素α4β7的配体,有助于将免疫抑制性T细胞亚群(Tr17细胞)保留在肠道内。这导致Tr17细胞转移到肿瘤和肿瘤引流淋巴结,从而损害免疫检查点阻断疗法。在接受免疫治疗的癌症患者中,低水平的血清可溶性MAdCAM-1与肠道菌群失调以及肾癌、膀胱癌和肺癌的不良临床结局相关。—Priscilla N. Kelly & Seth Thomas Scanlon
癌症对免疫检查点抑制剂(ICIs)的耐药性可能源于抗生素(ABX)治疗,这很可能是由于肠道微生物群发生偏离所致。当抗生素在ICI给药前(而非给药期间)使用时,会损害临床结果,这表明停用抗生素后的细菌重新定植可能是有害的。肠道共生菌会诱导FoxP3+视黄酸受体相关孤儿受体-γt(RORγt+)调节性(Treg17)细胞这一免疫抑制亚群的分化。在肠系膜淋巴结(mLNs)中启动或归巢至肠固有层的淋巴细胞表达α4β7整合素,该整合素与其反受体——粘膜地址素细胞粘附分子-1(MAdCAM-1)相互作用,MAdCAM-1在高内皮微静脉(HEVs)中表达。
我们假设,破坏能够保留Treg17细胞的MAdCAM-1–α4β7相互作用可能会导致它们从肠道迁移到肿瘤,从而损害ICI的抗癌效果。我们使用了两种互补的方法来观察肠道T细胞向外周皮下肿瘤和肿瘤引流淋巴结(tdLNs)的迁移:(i) 使用Kaede小鼠,其表达一种荧光蛋白,在回肠受到紫外线照射时发生光转换;(ii) 将羧基荧光素琥珀酰亚胺酯注射到mLNs中。此外,我们在肝脏中转基因强制表达Madcam1,以便在Treg17细胞迁移过程中局部截留它们。
几类抗生素下调了回肠微静脉、派尔集合淋巴结和mLNs中Madcam1的表达,同时伴随着表达α4β7的T辅助(Th17)细胞和Treg17细胞从回肠向肠外肿瘤和tdLNs的迁出。这种抗生素诱导的MAdCAM-1减少可以用肠梭菌属(包含梭状梭菌物种)在肠道的重新定植来解释,因为口服该菌属足以通过影响胆汁酸代谢来下调MAdCAM-1的表达。通过遗传或抗体介导的中和MAdCAM-1或α4β7整合素,可以模拟抗生素的免疫抑制效应,促进对靶向程序性细胞死亡蛋白1(PD-1)的ICIs的耐药性,并导致tdLNs和肿瘤中肠道来源的α4β7+ Treg17细胞激增。通过粪便微生物移植恢复回肠HEV上的MAdCAM-1或阻断IL-17A可以逆转抗生素的抑制作用。在肝脏中异位表达MAdCAM-1导致嗜肠道性的α4β7+ Treg17细胞在局部滞留,减少了它们在肿瘤床的积聚,并改善了小鼠的免疫治疗效果。最后,低血清可溶性MAdCAM-1被确定为肠道菌群失调的一个替代指标,并且是肾癌、膀胱癌和肺癌患者在接受靶向PD-1或PD-L1抗体的免疫治疗时总生存期和无进展生存期缩短的有力预测因子。在非小细胞肺癌患者中,可溶性MAdCAM-1的预后价值独立于PD-L1的表达。
嗜肠道性且具有免疫抑制作用的Treg17细胞向癌组织(肿瘤和tdLNs)的迁移,受到HEV地址素MAdCAM-1与Treg17细胞表达的整合素α4β7之间分子相互作用的抑制。抗生素或肠道菌群失调破坏MAdCAM-1的表达,会导致Treg17细胞迁移到肿瘤中,从而损害小鼠和患者的癌症免疫监视以及ICIs的治疗效果。
抗生素(ABX)会损害癌症患者中程序性细胞死亡蛋白1(PD-1)阻断疗法的疗效,但其免疫抑制作用的潜在机制仍不清楚。通过诱导回肠中粘膜地址素细胞粘附分子1(MAdCAM-1)的下调,抗生素后由肠梭菌属物种驱动的肠道重新定植,促使嗜肠道性的α4β7+CD4+调节性T17细胞迁移到肿瘤中。这些有害的抗生素效应可以通过口服肠梭菌属物种、遗传缺陷或抗体介导的中和MAdCAM-1及其受体α4β7整合素来模拟。相比之下,粪便微生物移植或白细胞介素-17A中和则能预防抗生素诱导的免疫抑制。在独立的肺癌、肾癌和膀胱癌患者队列中,低血清水平的可溶性MAdCAM-1具有负面的预后影响。因此,MAdCAM-1–α4β7轴构成了癌症免疫监视中一个可操作的肠道免疫检查点。
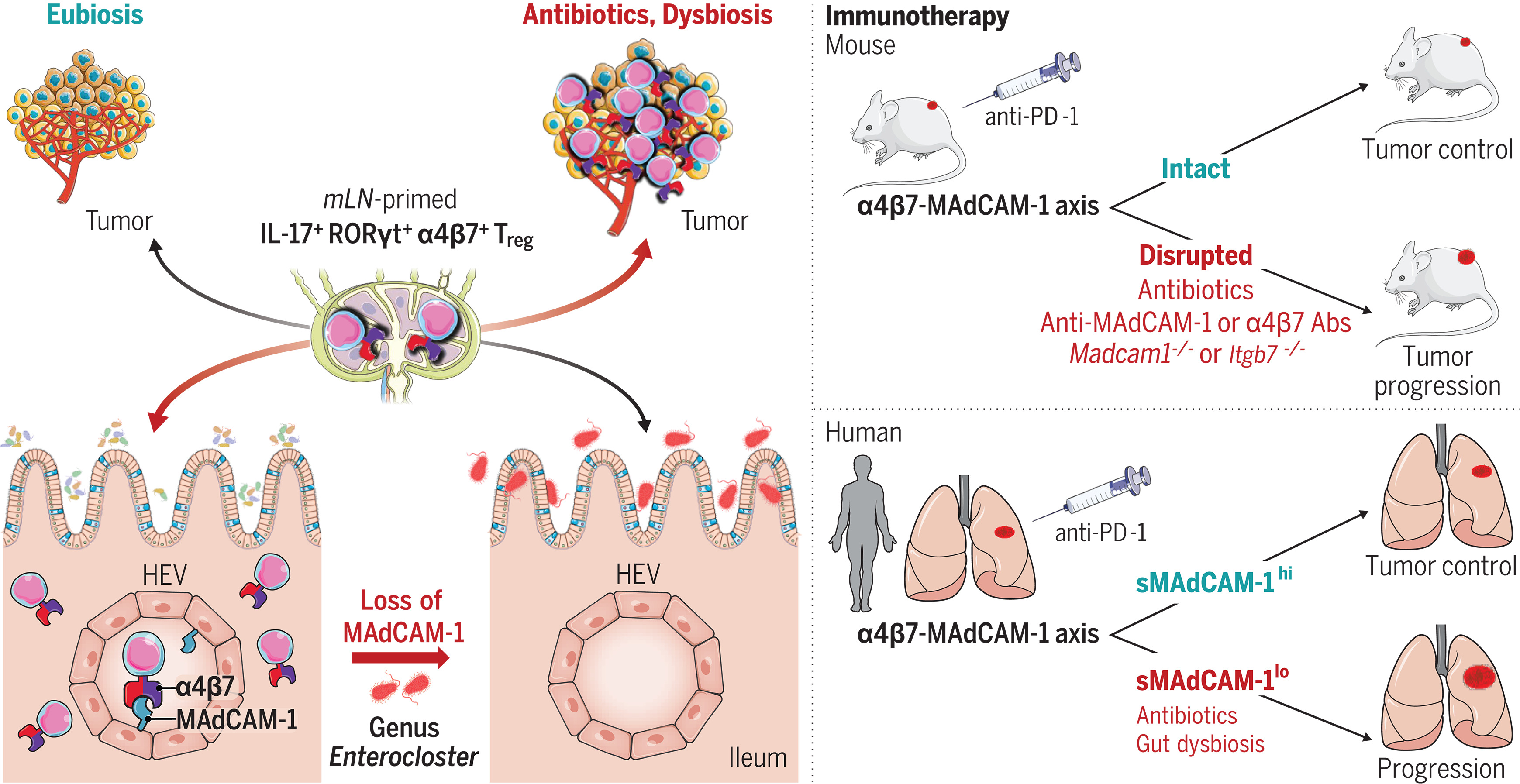
图:MAdCAM-1作为癌症免疫监视的肠道免疫检查点。例如,在停止抗生素治疗后,肠梭菌属细菌可诱导回肠固有层和肠系膜淋巴结中MAdCAM-1的下调,从而促使免疫抑制性α4β7+ Treg17细胞从肠道向外迁移至肿瘤组织及肿瘤引流淋巴结。破坏MAdCAM-1–α4β7轴会削弱小鼠及患者免疫治疗的疗效。
A microbiota-modulated checkpoint directs immunosuppressive intestinal T cells into cancers