Tertiary lymphoid structures (TLSs) are lymphoid formations that are found in nonlymphoid tissues. TLS can develop in inflamed tissues and are associated with chronic inflammatory disorders, autoimmunity, and cancer. In the setting of tumors, TLSs facilitate the influx of immune cells into the tumor site and have therefore attracted interest as a means of improving anticancer immunity and favorable treatment response in patients. Schumacher and Thommen review the biology of TLSs and outline recent advances in TLS research. They discuss how TLSs are detected and defined, the mechanism(s) of formation in cancer, and the potential of targeting TLSs for therapeutic benefit. —PNK
Tertiary lymphoid structures (TLSs) are organized aggregates of immune cells that form postnatally in nonlymphoid tissues. TLSs are not found under physiological conditions but arise in the context of chronic inflammation, such as in autoimmune disease, chronic infection, and cancer. With few exceptions, the presence of TLSs in tumors correlates with better prognosis and clinical outcome upon immunotherapy, but, in spite of their presumed importance, the drivers of TLS formation in cancer and the contribution of these structures to intratumoral immune responses remain incompletely understood.
TLSs resemble secondary lymphoid organs (SLOs) anatomically, and it was originally assumed that their formation would largely be induced by the same stimuli. However, the cell pools and signals that provide inductive stimuli for TLS formation are at least partially different. For instance, several observations suggest that tumor-specific T and B cell immunity may induce some of the molecular factors required for TLS formation and maintenance, and heterogeneity in these drivers may result in distinct TLS states.
It has been speculated that TLSs recapitulate SLO functions at the inflamed tissue site, and available evidence suggests that a contribution of TLSs to the strength of tumor-specific immune responses is plausible. However, whether such a contribution primarily involves the boosting of T cell responses generated in SLOs or the development of new T and B cell reactivities remains a key unanswered question. In addition, the presence of TLSs at the tumor site may offer the possibility for the generation of qualitatively distinct immune responses. Specifically, because TLSs are not encapsulated, exposure of TLS-resident immune cells to macromolecules from the inflamed microenvironment appears to be a realistic possibility, and this could potentially sculpt the nature of intratumoral immune responses. Finally, recent studies suggest a role for TLSs in the clinical response to immune checkpoint blockade, which may make these structures attractive therapeutic targets. However, the development of such strategies should take into account the possible consequences of ectopic formation of lymphoid tissue at other body sites.
The prognostic and predictive value of TLSs in cancer has strengthened the interest in these structures as potential mediators of antitumor immunity. Although TLSs have been identified in many cancer types, the markers used to define and characterize TLSs have often varied across studies, complicating efforts to compare predictive value and to assess TLS heterogeneity between cancer types. Thus, the development of standardized approaches to measure TLS number and composition is likely to further reveal their predictive and prognostic value in different disease settings. Related to this, a more comprehensive characterization of TLSs may potentially lead to the identification of a spectrum of TLS states, based on aspects such as cellular composition, location, maturation, and function. Similar to the definition of T cell states in cancer, which has substantially improved our understanding of the role of specific T cell populations in tumor-specific immunity, the molecular definition of TLS states may help to improve their value as prognostic and predictive markers. Finally, a better appreciation of TLS function and the potential contribution of TLSs to autoimmune toxicity will be important to maximize their value as therapeutic targets.
Ectopic lymphoid aggregates, termed tertiary lymphoid structures (TLSs), are formed in numerous cancer types, and, with few exceptions, their presence is associated with superior prognosis and response to immunotherapy. In spite of their presumed importance, the triggers that lead to TLS formation in cancer tissue and the contribution of these structures to intratumoral immune responses remain incompletely understood. Here, we discuss the present knowledge on TLSs in cancer, focusing on (i) the drivers of TLS formation, (ii) the function and contribution of TLSs to the antitumor immune response, and (iii) the potential of TLSs as therapeutic targets in human cancers.
三级淋巴结构是发现于非淋巴组织中的淋巴组织结构。TLS可在炎症组织中形成,并与慢性炎症性疾病、自身免疫和癌症相关。在肿瘤环境中,TLS促进了免疫细胞向肿瘤部位的聚集,因此作为改善患者抗癌免疫和有利治疗反应的一种手段而引起了关注。Schumacher和Thommen回顾了TLS的生物学特性,并概述了TLS研究的最新进展。他们讨论了如何检测和定义TLS、其在癌症中的形成机制以及靶向TLS以获得治疗益处的潜力。—PNK
三级淋巴结构是在非淋巴组织中出生后形成的免疫细胞有组织聚集体。在生理条件下并不存在TLS,它们出现在慢性炎症的背景下,例如自身免疫性疾病、慢性感染和癌症。除少数例外,肿瘤中TLS的存在与更好的预后和免疫治疗后的临床结果相关,但尽管它们被认为很重要,癌症中TLS形成的驱动因素以及这些结构对瘤内免疫反应的贡献仍未完全明了。
TLS在解剖结构上类似于次级淋巴器官,最初假设其形成主要由相同的刺激诱导。然而,为TLS形成提供诱导刺激的细胞池和信号至少部分不同。例如,一些观察表明,肿瘤特异性T细胞和B细胞免疫可能诱导TLS形成和维持所需的一些分子因子,这些驱动因素的异质性可能导致不同的TLS状态。
据推测,TLS在炎症组织部位重演了SLO的功能,现有证据表明TLS对肿瘤特异性免疫反应强度的贡献是合理的。然而,这种贡献主要涉及增强在SLO中产生的T细胞反应,还是涉及新的T细胞和B细胞反应性的产生,仍然是一个关键的未解问题。此外,肿瘤部位存在TLS可能为产生性质上独特的免疫反应提供了可能性。具体来说,由于TLS没有包膜,TLS驻留的免疫细胞暴露于炎症微环境中的大分子似乎是现实的可能性,这可能会塑造瘤内免疫反应的性质。最后,最近的研究表明TLS在免疫检查点阻断的临床反应中起作用,这可能使这些结构成为有吸引力的治疗靶点。然而,制定此类策略时应考虑在其他身体部位异位形成淋巴组织的可能后果。
TLS在癌症中的预后和预测价值增强了人们对这些结构作为抗肿瘤免疫潜在介导因子的兴趣。尽管已在许多癌症类型中发现了TLS,但用于定义和表征TLS的标记物在不同研究中往往各不相同,这使比较预测价值和评估癌症类型间TLS异质性的工作变得复杂。因此,开发测量TLS数量和组成的标准化方法可能会进一步揭示它们在不同疾病环境中的预测和预后价值。与此相关的是,更全面地描述TLS特征,可能基于细胞组成、位置、成熟度和功能等方面,识别出一系列TLS状态。类似于癌症中T细胞状态的定义极大地增进了我们对特定T细胞群在肿瘤特异性免疫中作用的理解,TLS状态的分子定义可能有助于提高其作为预后和预测标记物的价值。最后,更好地认识TLS功能以及TLS对自身免疫毒性的潜在贡献,对于最大化其作为治疗靶点的价值至关重要。
在许多癌症类型中都会形成称为三级淋巴结构的异位淋巴聚集体,除少数例外,它们的存在与更好的预后和对免疫治疗的反应相关。尽管它们被认为很重要,但导致癌组织中TLS形成的触发因素以及这些结构对瘤内免疫反应的贡献仍未完全明了。在此,我们讨论了目前关于癌症中TLS的知识,重点关注(i)TLS形成的驱动因素,(ii)TLS的功能及其对抗肿瘤免疫反应的贡献,以及(iii)TLS作为人类癌症治疗靶点的潜力。
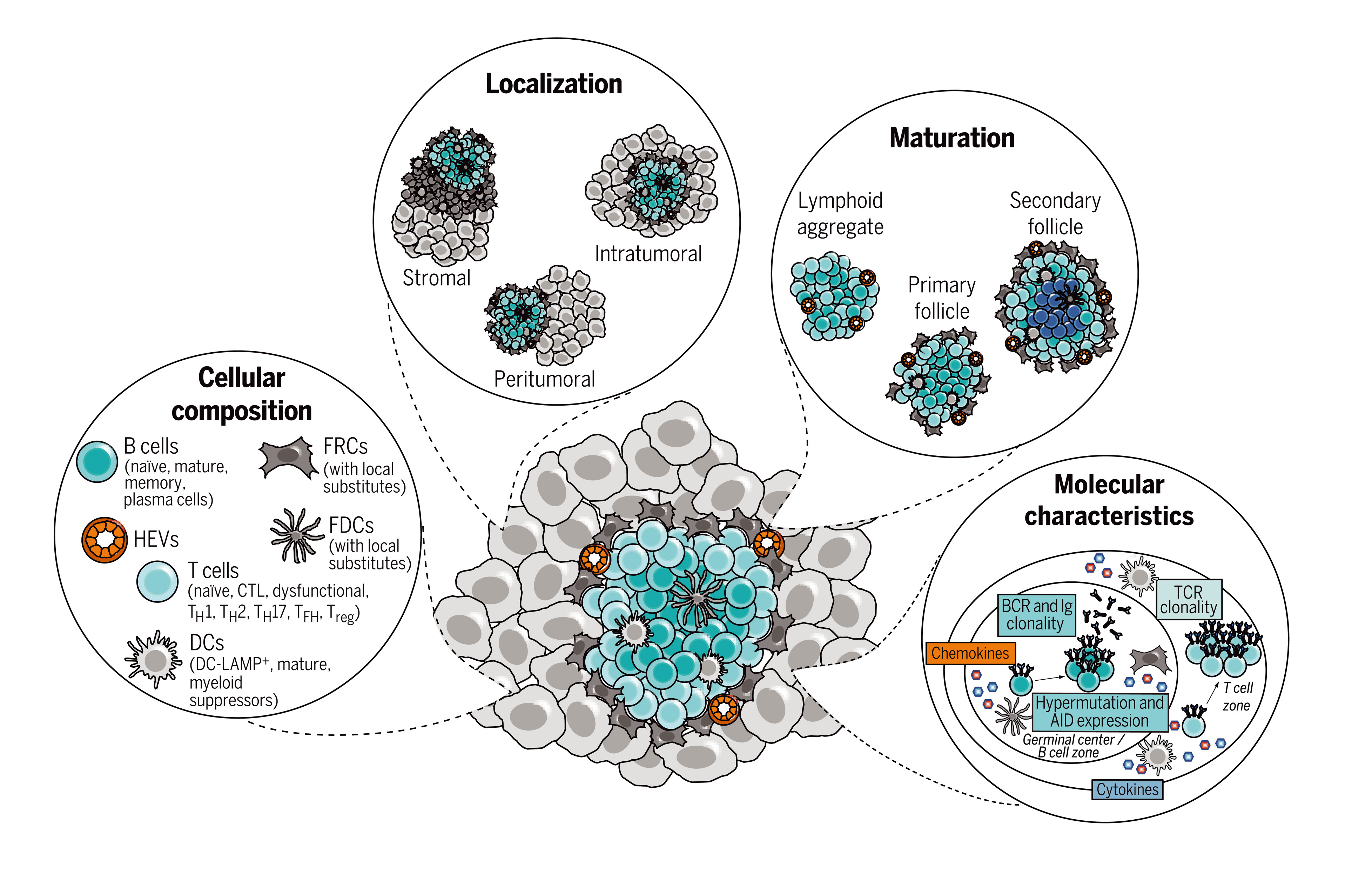
图:定义TLS状态。TLS状态的分子定义可能推动其作为预后和预测标志物的应用。能够揭示TLS状态多样性的特征包括其细胞组成、位置和成熟度;其细胞因子和趋化因子环境的特性;以及其B细胞受体(BCR)和T细胞受体(TCR)库。AID,激活诱导胞苷脱氨酶;CTL,细胞毒性T淋巴细胞;FRCs,滤泡网状细胞;FDCs,滤泡树突状细胞;HEVs,高内皮微静脉;Ig,免疫球蛋白;TFH,滤泡辅助性T细胞;TH,辅助性T细胞;Treg,调节性T细胞。