The tumor microenvironment contains many different kinds of immune cells, the composition, function, and roles of which are unclear. Using single-cell RNA sequencing of T cells in 21 cancer types from more than 300 patients, Zheng et al. identified differences in transcript composition that could be used to catalog different T cell types (see the Perspective by van der Leun and Schumacher). These annotations identified the different roles of specific types of CD4+ and CD8+ T cells among the different tumor types. Some of these clusters revealed evidence for two developmental paths for T cells, one of which shows a trajectory toward the “exhausted” T cell state, knowledge of which may be useful in developing future cancer immunotherapies. —LMZ
Cancer immunotherapies that target tumor-specific T cells have benefited many cancer patients, but the clinical efficacy varies greatly among different cancer types. Tumor-infiltrating T cells often enter a dysfunctional state, widely known as T cell exhaustion, and the antitumor functions of effector T cells are regulated by multiple factors, including the presence of regulatory T cells (Treg cells). The states and abundances of T cells vary across tumor microenvironments (TMEs) of different cancer types, which may fundamentally influence different clinical parameters such as drug response to immunotherapies.
To build a high-resolution pan-cancer T cell atlas, we performed single-cell RNA sequencing (scRNA-seq) on tumors, paracancerous tissues, and blood samples from patients of various cancer types and collected additional published scRNA-seq datasets. The diverse data were integrated after correcting confounding factors and batch effects. This atlas was composed of scRNA-seq data from 316 patients across 21 cancer types. T cell receptor (TCR) sequences of individual T cells with gene expression profiles were assembled to characterize the expansion and dynamics of T cells. Various computational methods were applied to investigate the features and abundance of T cells across cancer types.
We identified multiple potentially tumor-reactive T cell (pTRT) populations in cancer patients. The states of the pTRTs varied dramatically in the tumor microenvironment of different cancer types. For CD8+ T cells, the major pTRTs were exhausted T cells and exhibited high heterogeneity. We computationally inferred two major developmental paths to T cell exhaustion, through effector memory T cells and tissue-resident memory T cells, respectively, and both were prevalent among cancer types. We also noted the state transitions between terminal exhausted T cells and cells such as natural killer (NK)–like T cells, Type 17 CD8+ T cells (Tc17 cells) cells, and CD8+ Treg cells, but such transitions tend to occur in specific cancer types. For CD4+ T cells, follicular helper T cell (TFH)/T helper 1 (TH1) dual-functional T cells, which appeared to originate from TFH cells, were also notable pTRTs and correlated with the tumor mutation burden. We also found that the transcriptional programs of pTRTs could be affected by transforming growth factor–β (TGF-β) and interferons in the TMEs. The abundances of T cell states vary dramatically depending on cancer types. On the basis of tumor-infiltrating T cell compositions, cancer patients could be immune-typed as a group with high frequencies of terminal exhausted CD8+ T cells and another group with high frequencies of tissue-resident memory CD8+ T cells, and the immune types were associated with clinical traits such as patient survival and responses to immune checkpoint blockade.
We depicted the pan-cancer landscape of T cell heterogeneity and dynamics in the TME and established a baseline reference for future temporal or spatial studies associated with cancer treatments. The systematic comparison across cancer types revealed the commonalities and differences of T cell states in different TMEs. Our detailed signature, dynamics, and regulations of tumor-infiltrating T cells will facilitate the development of immunotherapies, and our proposed immune-typing can aid the therapeutic and diagnostic strategies that target T cells.
T cells play a central role in cancer immunotherapy, but we lack systematic comparison of the heterogeneity and dynamics of tumor-infiltrating T cells across cancer types. We built a single-cell RNA-sequencing pan-cancer atlas of T cells for 316 donors across 21 cancer types and revealed distinct T cell composition patterns. We found multiple state-transition paths in the exhaustion of CD8+ T cells and the preference of those paths among different tumor types. Certain T cell populations showed specific correlation with patient properties such as mutation burden, shedding light on the possible determinants of the tumor microenvironment. T cell compositions within tumors alone could classify cancer patients into groups with clinical trait specificity, providing new insights into T cell immunity and precision immunotherapy targeting T cells.
肿瘤微环境中包含许多不同种类的免疫细胞,其组成、功能和作用尚不清楚。通过对来自300多名患者、21种癌症类型的T细胞进行单细胞RNA测序,Zheng等人识别出了可用于分类不同T细胞类型的转录组组成差异(参见van der Leun和Schumacher的观点文章)。这些注释明确了特定类型的CD4+和CD8+ T细胞在不同肿瘤类型中的不同作用。其中一些细胞群揭示了T细胞的两条发育路径证据,其中一条显示其轨迹趋向于“耗竭”T细胞状态,了解这一点可能对开发未来的癌症免疫疗法有所帮助。—LMZ
针对肿瘤特异性T细胞的癌症免疫疗法已使许多癌症患者受益,但不同癌症类型的临床疗效差异很大。肿瘤浸润性T细胞常进入一种功能障碍状态,即广为人知的T细胞耗竭,而效应T细胞的抗肿瘤功能受到多种因素调控,包括调节性T细胞(Treg细胞)的存在。T细胞的状态和丰度在不同癌症类型的肿瘤微环境中各不相同,这可能从根本上影响不同的临床参数,例如对免疫疗法的药物反应。
为了构建高分辨率的泛癌T细胞图谱,我们对多种癌症类型患者的肿瘤组织、癌旁组织和血液样本进行了单细胞RNA测序,并收集了其他已发表的scRNA-seq数据集。在校正混杂因素和批次效应后,整合了这些多样化数据。该图谱包含来自21种癌症类型、316名患者的scRNA-seq数据。组装了具有基因表达谱的单个T细胞的T细胞受体序列,以表征T细胞的扩增和动态变化。应用多种计算方法研究了跨癌症类型的T细胞特征和丰度。
我们在癌症患者中鉴定出多个潜在的肿瘤反应性T细胞群体。pTRT的状态在不同癌症类型的肿瘤微环境中差异巨大。对于CD8+ T细胞,主要的pTRT是耗竭性T细胞,并表现出高度异质性。我们通过计算推断出T细胞耗竭的两条主要发育路径,分别通过效应记忆T细胞和组织驻留记忆T细胞,且这两条路径在各类癌症中都很普遍。我们还注意到终末耗竭T细胞与自然杀伤样T细胞、17型CD8+ T细胞(Tc17细胞)和CD8+ Treg细胞等之间的状态转换,但此类转换往往发生在特定的癌症类型中。对于CD4+ T细胞,看起来起源于滤泡辅助性T细胞的滤泡辅助性T细胞/T辅助1细胞双功能T细胞也是显著的pTRT,并与肿瘤突变负荷相关。我们还发现,pTRT的转录程序可能受到TME中转化生长因子-β和干扰素的影响。T细胞状态的丰度因癌症类型不同而差异巨大。基于肿瘤浸润T细胞的组成,癌症患者可以被免疫分型为一组具有高频率终末耗竭CD8+ T细胞,另一组具有高频率组织驻留记忆CD8+ T细胞,并且这些免疫类型与患者的生存和对免疫检查点阻断的反应等临床特征相关。
我们描绘了TME中T细胞异质性和动态的泛癌图谱,并为未来与癌症治疗相关的时间或空间研究建立了基线参考。跨癌症类型的系统比较揭示了不同TME中T细胞状态的共性与差异。我们关于肿瘤浸润T细胞的详细特征、动态和调控机制将促进免疫疗法的发展,而我们提出的免疫分型有助于制定针对T细胞的治疗和诊断策略。
T细胞在癌症免疫治疗中起着核心作用,但我们缺乏对不同癌症类型中肿瘤浸润T细胞的异质性和动态性的系统比较。我们为21种癌症类型的316名供体构建了T细胞的单细胞RNA测序泛癌图谱,并揭示了不同的T细胞组成模式。我们发现了CD8+ T细胞耗竭过程中的多条状态转换路径,以及这些路径在不同肿瘤类型中的倾向性。某些T细胞群体与患者的突变负荷等特性表现出特定的相关性,为肿瘤微环境的可能决定因素提供了线索。仅凭肿瘤内的T细胞组成即可将癌症患者分类为具有临床特征特异性的组别,这为T细胞免疫和针对T细胞的精准免疫治疗提供了新见解。
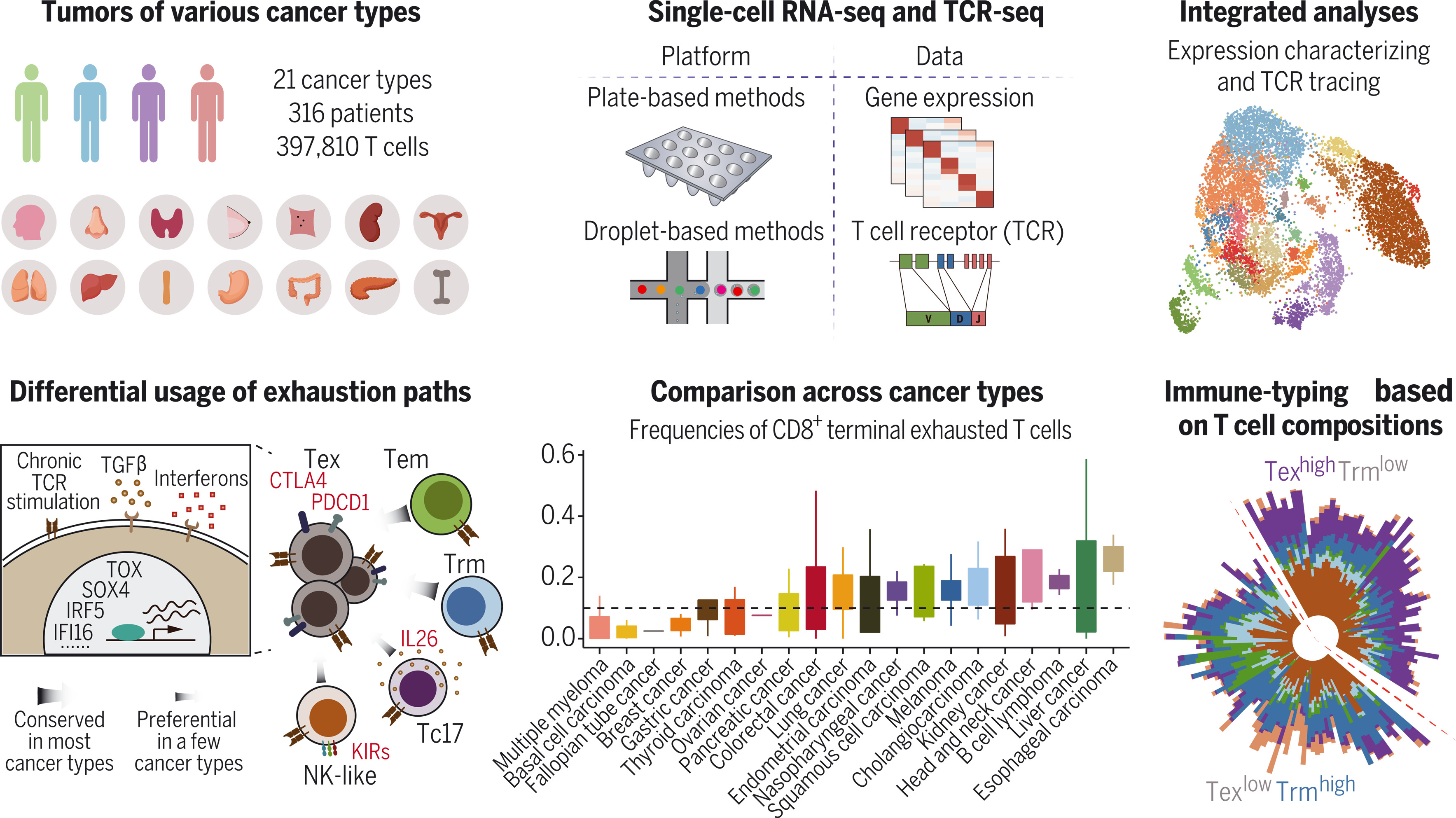
图:人类泛癌症T细胞图谱的系统分析.我们通过单细胞RNA测序技术分析了来自21种癌症类型、316名患者的约39万个T细胞。结合基因表达谱与T细胞受体序列,我们探究了肿瘤浸润性T细胞的异质性和动态变化,并对不同癌症类型的T细胞进行了系统性比较。此外,我们提出了一种基于T细胞组成的免疫分型方案。KIR:杀伤细胞免疫球蛋白样受体;IL26:白细胞介素-26。
Pan-cancer single-cell landscape of tumor-infiltrating T cells