Therapeutic interventions are needed for prostate tumors that exhibit a loss of androgen receptor dependence in castration-resistant prostate cancer (CRPC). However, there is a scarcity of prostate cancer cell line models. Tang et al. performed a molecular study on a number of CRPC organoids, cell lines, and patient-derived xenographs by combining sequencing techniques and transcriptomics to identify four distinct genetic subgroups of tumors. Dominant transcription factors were identified for each subgroup, with a deeper analysis of subgroup 4 leading to the identification of a chromatin-opening positive feedback loop. From these data, the authors propose that inhibitors of the transcriptional coactivators YAP and TAZ may be used to treat individuals with subgroup 4–like tumors, the second most common type of CRPC observed in this study. —LMZ
Untreated prostate cancers rely on androgen receptor (AR) signaling for growth and survival, forming the basis for the initial efficacy of androgen deprivation therapy (ADT). Yet the disease can relapse and progress to a lethal stage termed castration-resistant prostate cancer (CRPC). Reactivation of AR signaling represents the most common driver of CRPC growth, and next-generation AR signaling inhibitors (ARSIs) are now used in combination with ADT as a first-line therapy. However, ARSIs can result in selective pressure, thereby generating AR-independent tumors. The transition from AR dependence frequently accompanies a change in phenotype resembling developmental transdifferentiation or “lineage plasticity.” Neuroendocrine prostate cancer, which lacks a defined pathologic classification, is the most studied type of lineage plasticity. However, most AR-null tumors do not exhibit neuroendocrine features and are classified as “double-negative prostate cancer,” the drivers of which are poorly defined.
Lineage plasticity studies in CRPC are limited by the lack of genetically defined patient-derived models that recapitulate the disease spectrum. To address this, we developed a biobank of organoids generated from patient biopsies to study the landscape of metastatic CRPC and allow for functional validation assays. Proteins called transcription factors (TFs) are drivers of tumor lineage plasticity. To identify the key TFs that drive the growth of AR-independent tumors, we integrated epigenetic and transcriptomic data generated from CRPC models.
We generated ATAC-seq (assay for transposase-accessible chromatin sequencing) and RNA-seq data from 22 metastatic human prostate cancer organoids, six patient-derived xenografts (PDXs), and 12 derived or traditional cell lines. We classified the 40 models into four subtypes and predicted key TFs of each subtype. We identified the well-characterized AR-dependent (CRPC-AR) and neuroendocrine subtypes (CRPC-NE) as well as two AR-negative/low groups, including a Wnt-dependent subtype (CRPC-WNT), driven by TCF/LEF TFs, and a stem cell–like (SCL) subtype (CRPC-SCL), driven by the AP-1 family of TFs. We applied RNA-seq signatures derived from the organoids to 366 patient samples from two independent CRPC datasets, which recapitulated the four-subtype classification. We found that CRPC-SCL is the second most prevalent group and is associated with shorter time under ARSI treatment compared to CRPC-AR. Additional chromatin immunoprecipitation sequencing (ChIP-seq) analysis indicated that AP-1 works together with the proteins YAP, TAZ, and TEAD, revealing YAP/TAZ and AP-1 as potential actionable targets in CRPC-SCL. Using overexpression assays in AR-high cells, we revealed how AP-1 functions as a pioneering factor and master regulator for CRPC-SCL.
By using a diverse biobank of organoids, PDXs, and cell lines that recapitulate the heterogeneity of metastatic prostate cancer, we created a map of the chromatin accessibility and transcriptomic landscape of CRPC. We validated the CRPC-AR and CRPC-NE subtypes and report two subtypes of AR-negative/low samples as well as their respective key TFs. Additional analysis revealed a model in which YAP, TAZ, TEAD, and AP-1 function together and drive oncogenic growth in CRPC-SCL samples. Overall, our results show how stratification of CRPC patients into four subtypes using their transcriptomes can potentially inform appropriate clinical decisions.
In castration-resistant prostate cancer (CRPC), the loss of androgen receptor (AR) dependence leads to clinically aggressive tumors with few therapeutic options. We used ATAC-seq (assay for transposase-accessible chromatin sequencing), RNA-seq, and DNA sequencing to investigate 22 organoids, six patient-derived xenografts, and 12 cell lines. We identified the well-characterized AR-dependent and neuroendocrine subtypes, as well as two AR-negative/low groups: a Wnt-dependent subtype, and a stem cell–like (SCL) subtype driven by activator protein–1 (AP-1) transcription factors. We used transcriptomic signatures to classify 366 patients, which showed that SCL is the second most common subtype of CRPC after AR-dependent. Our data suggest that AP-1 interacts with the YAP/TAZ and TEAD proteins to maintain subtype-specific chromatin accessibility and transcriptomic landscapes in this group. Together, this molecular classification reveals drug targets and can potentially guide therapeutic decisions.
对于在去势抵抗性前列腺癌(CRPC)中表现出雄激素受体依赖性丧失的前列腺肿瘤,需要进行治疗干预。然而,前列腺癌细胞系模型十分缺乏。Tang等人通过对多个CRPC类器官、细胞系和患者来源的异种移植模型进行分子研究,结合测序技术和转录组学分析,确定了四种不同的肿瘤遗传亚型。研究为每个亚型鉴定了主导的转录因子,并对亚型4进行了更深入的分析,从而鉴定出一个染色质开放的正反馈回路。基于这些数据,作者提出转录共激活因子YAP和TAZ的抑制剂或可用于治疗具有类似亚型4肿瘤的个体,该亚型是本研究观察到的第二常见的CRPC类型。 —LMZ
未经治疗的前列腺癌依赖雄激素受体(AR)信号传导来维持生长和存活,这构成了雄激素剥夺疗法(ADT)初始有效性的基础。然而,该疾病可能复发并进展至一个称为去势抵抗性前列腺癌(CRPC)的致死阶段。AR信号传导的重新激活是CRPC生长最常见的驱动因素,新一代AR信号抑制剂(ARSIs)现与ADT联合用作一线疗法。然而,ARSIs可能导致选择性压力,从而产生不依赖AR的肿瘤。从AR依赖性的转变常常伴随着表型的变化,类似于发育中的转分化或“谱系可塑性”。神经内分泌前列腺癌是研究最深入的谱系可塑性类型,但其缺乏明确的病理学分类。然而,大多数AR缺失的肿瘤并不表现出神经内分泌特征,被归类为“双阴性前列腺癌”,其驱动因素尚不明确。
CRPC中的谱系可塑性研究受限于缺乏能够概括疾病谱系的、遗传学定义明确的患者来源模型。为解决这一问题,我们建立了一个来源于患者活检组织的类器官生物库,用于研究转移性CRPC的全貌并进行功能验证实验。被称为转录因子(TFs)的蛋白质是肿瘤谱系可塑性的驱动因素。为了鉴定驱动AR非依赖性肿瘤生长的关键TFs,我们整合了从CRPC模型生成的表观遗传和转录组数据。
我们从22个转移性人类前列腺癌类器官、6个患者来源的异种移植模型(PDXs)和12个衍生或传统细胞系生成了ATAC-seq(转座酶可及染色质测序)和RNA-seq数据。我们将这40个模型分为四种亚型,并预测了每个亚型的关键TFs。我们鉴定了特征明确的AR依赖性亚型(CRPC-AR)和神经内分泌亚型(CRPC-NE),以及两个AR阴性/低表达组:一个是由TCF/LEF TFs驱动的Wnt依赖性亚型(CRPC-WNT),另一个是由AP-1家族TFs驱动的干细胞样(SCL)亚型(CRPC-SCL)。我们将从类器官中获得的RNA-seq特征应用于来自两个独立CRPC数据集的366个患者样本,重现了这四种亚型的分类。我们发现,与CRPC-AR相比,CRPC-SCL是第二普遍的组别,并且与接受ARSI治疗时间较短相关。额外的染色质免疫沉淀测序(ChIP-seq)分析表明,AP-1与YAP、TAZ和TEAD蛋白协同作用,揭示了YAP/TAZ和AP-1是CRPC-SCL中潜在的可操作靶点。通过在AR高表达细胞中使用过表达实验,我们揭示了AP-1如何作为CRPC-SCL的先导因子和主调控因子发挥作用。
通过利用一个能概括转移性前列腺癌异质性的、多样化的类器官、PDXs和细胞系生物库,我们绘制了CRPC的染色质可及性和转录组图谱。我们验证了CRPC-AR和CRPC-NE亚型,并报告了AR阴性/低样本的两种亚型及其各自的关键TFs。进一步分析揭示了一个模型,其中YAP、TAZ、TEAD和AP-1共同作用,驱动CRPC-SCL样本中的致癌性生长。总的来说,我们的结果表明,利用转录组将CRPC患者分为四种亚型如何可能为适当的临床决策提供信息。
在去势抵抗性前列腺癌(CRPC)中,雄激素受体(AR)依赖性的丧失导致了临床上具有侵袭性且治疗选择极少的肿瘤。我们使用ATAC-seq(转座酶可及染色质测序)、RNA-seq和DNA测序研究了22个类器官、6个患者来源的异种移植模型和12个细胞系。我们鉴定了特征明确的AR依赖性和神经内分泌亚型,以及两个AR阴性/低表达组:一个Wnt依赖性亚型,和一个由激活蛋白-1(AP-1)转录因子驱动的干细胞样(SCL)亚型。我们利用转录组特征对366名患者进行了分类,结果显示SCL是继AR依赖性之后第二常见的CRPC亚型。我们的数据表明,在该组中,AP-1与YAP/TAZ和TEAD蛋白相互作用,以维持亚型特异性的染色质可及性和转录组景观。总之,这种分子分类揭示了药物靶点,并可能指导治疗决策。
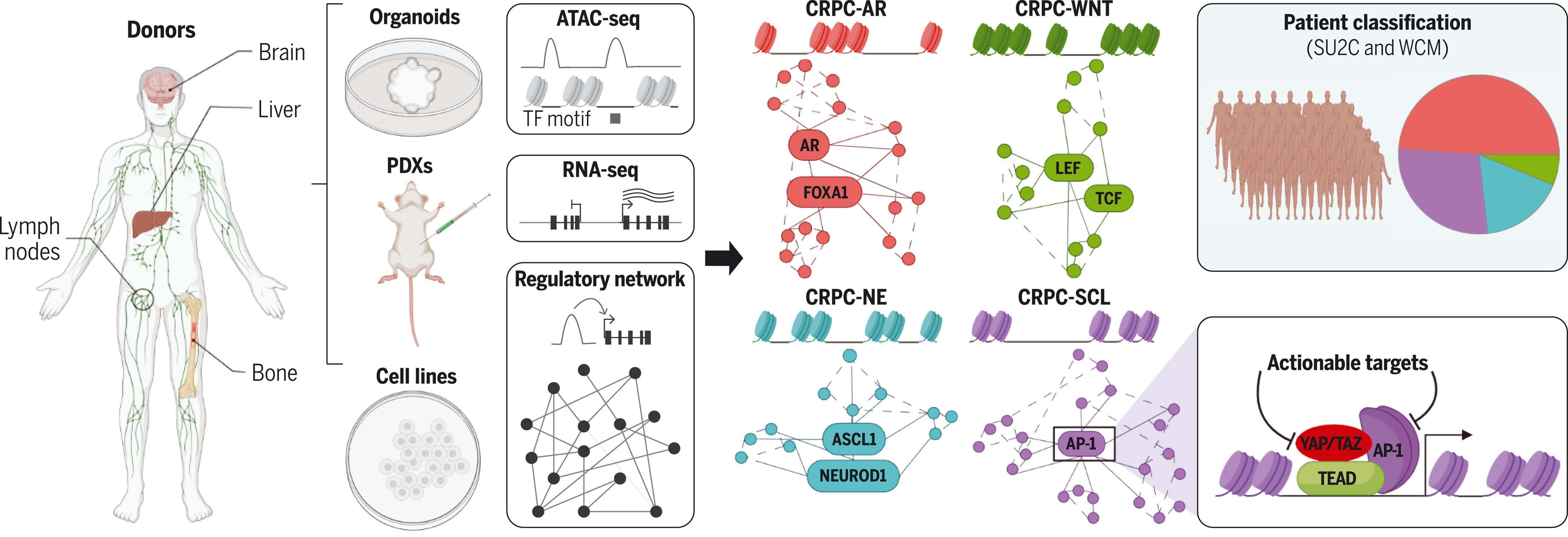
图:通过整合类器官、患者来源异种移植物及细胞系的染色质可及性与转录组数据,鉴定出去势抵抗性前列腺癌的四种亚型。TF,转录因子;AR,雄激素受体;NE,神经内分泌型;SCL,干细胞样型。YAP/TAZ/TEAD/AP-1在CRPC-SCL中的协同作用提示了可干预的靶点。将模型衍生的RNA-seq特征应用于366例患者样本,成功复现了这四种亚型的分类。
Chromatin profiles classify castration-resistant prostate cancers suggesting therapeutic targets