Historical accounts linking cancer and microbes date as early as four millennia ago. After establishment of the germ theory of infectious diseases, clinical research of microbial influences on cancer began in 1868, when William Busch reported spontaneous tumor regressions in patients with Streptococcus pyogenes infections. Over the next century, poor reproducibility, erroneous microbiological claims, and severe toxicity led many to discount the role of bacteria in carcinogenesis and cancer therapy. However, these studies provided the first crude demonstrations of cancer immunotherapy. Contemporaneously, the viral theory of cancer flourished, spurred by the 1911 discovery of Rous sarcoma virus, which transformed benign tissue into malignant tumors in chickens. The decades-long search to find viruses behind every human cancer ultimately failed, and many cancers have been linked to somatic mutations. Now the field is encountering intriguing claims of the importance of microbes, including bacteria and fungi, in cancer and cancer therapy. This Review critically evaluates this evidence in light of modern cancer biology and immunology, delineating roles for microbes in cancer by examining advances in proposed mechanisms, diagnostics, and modulation strategies.
Few microbes directly cause cancer, but many seem complicit in its growth, often acting through the host’s immune system; conversely, several have immunostimulatory properties. Mechanistic analyses of gut microbiota–immune system interactions reveal powerful effects on antitumor immunity by modulating primary and secondary lymphoid tissue activities. Many of these pathways invoke Toll-like receptor–initiated cytokine signaling, but microbial metabolic effects and antigenic mimicry with cancer cells are also important. In preclinical models, microbial metabolites also regulate phenotypes of tumor somatic mutations and modulate immune checkpoint inhibitor efficacy.
Emerging evidence suggests that intratumoral bacteria exist and are active, with overlapping immunohistochemistry, immunofluorescence, electron microscopy, and sequencing data in ~10 cancer types. Preliminary studies further suggest that fungi and bacteriophages contribute to gastrointestinal cancers. However, the abundance of intratumoral microbial cells is low relative to cancer cells, and knowledge of their functional repertoire and potency remains limited. Further validation of their prevalence and impact is needed in diverse cohorts and therapeutic contexts.
The immunomodulatory effects of host microbiota have reinvigorated efforts to change their composition as a form of immunotherapy. Despite extensive preclinical evidence, translation of microbiota modulation approaches into humans has not yet materialized into commercialized therapies. Synthetic biology approaches are also gaining traction, with engineered bacterial cancer therapies in preclinical and clinical trial settings.
The prognostic and predictive value of TLSs in cancer has strengthened the interest in these structures as potential mediators of antitumor immunity. Although TLSs have been identified in many cancer types, the markers used to define and characterize TLSs have often varied across studies, complicating efforts to compare predictive value and to assess TLS heterogeneity between cancer types. Thus, the development of standardized approaches to measure TLS number and composition is likely to further reveal their predictive and prognostic value in different disease settings. Related to this, a more comprehensive characterization of TLSs may potentially lead to the identification of a spectrum of TLS states, based on aspects such as cellular composition, location, maturation, and function. Similar to the definition of T cell states in cancer, which has substantially improved our understanding of the role of specific T cell populations in tumor-specific immunity, the molecular definition of TLS states may help to improve their value as prognostic and predictive markers. Finally, a better appreciation of TLS function and the potential contribution of TLSs to autoimmune toxicity will be important to maximize their value as therapeutic targets.
A better understanding of the roles of microbes in cancer provides an opportunity to improve each stage of the cancer care cycle, but major challenges remain. Concerted efforts to characterize cancer-associated microbiota among tumor, stool, and blood samples with gold-standard contamination controls would tremendously aid this progress. This would be analogous to The Cancer Genome Atlas’s role in characterizing the cancer somatic mutation landscape. Large-scale clinical trials are currently testing the efficacy of microbiota modulation approaches, ranging from dietary modifications to intratumorally injected, engineered bacteria. These bacterial cancer therapies, if safe and effective, could tremendously expand the cancer therapy armamentarium. Altogether, integrating the host-centric and microbial viewpoints of cancer may improve patient outcomes while providing a nuanced understanding of cancer-host-microbial evolution.
将癌症与微生物联系起来的历史记载可追溯到四千年前。在传染病细菌理论确立后,关于微生物对癌症影响的临床研究始于1868年,当时威廉·布施报道了化脓性链球菌感染患者中出现的自发性肿瘤消退。在接下来的一个世纪里,重复性差、错误的微生物学主张以及严重的毒性反应,导致许多人否定了细菌在癌变和癌症治疗中的作用。然而,这些研究提供了癌症免疫疗法的首次粗略演示。与此同时,癌症的病毒理论蓬勃发展,这得益于1911年发现的劳斯肉瘤病毒,该病毒能使鸡的良性组织转化为恶性肿瘤。长达数十年寻找导致每种人类癌症的病毒的努力最终失败了,并且许多癌症已被证实与体细胞突变有关。如今,该领域正面临一些引人关注的主张,认为微生物(包括细菌和真菌)在癌症及癌症治疗中具有重要性。本综述结合现代癌症生物学和免疫学知识,通过审视所提出机制、诊断学和调控策略方面的进展,批判性地评估这些证据,从而阐明微生物在癌症中的作用。
直接导致癌症的微生物很少,但许多微生物似乎参与了癌症的生长,通常通过宿主的免疫系统发挥作用;相反,有些微生物具有免疫刺激特性。对肠道微生物群与免疫系统相互作用的机制分析揭示,通过调节初级和次级淋巴组织的活动,微生物对抗肿瘤免疫力产生了强大影响。其中许多通路涉及Toll样受体启动的细胞因子信号传导,但微生物的代谢效应以及与癌细胞的抗原模拟也很重要。在临床前模型中,微生物代谢物也调节肿瘤体细胞突变的表型,并影响免疫检查点抑制剂的疗效。
新出现的证据表明,肿瘤内细菌存在且具有活性,在大约10种癌症类型中,免疫组化、免疫荧光、电镜和测序数据存在重叠。初步研究进一步表明,真菌和噬菌体与胃肠道癌症有关。然而,相对于癌细胞,肿瘤内微生物细胞的丰度较低,对其功能谱和效力的了解仍然有限。需要在不同的队列和治疗背景下进一步验证其普遍性和影响。
宿主微生物群的免疫调节作用重新激发了人们改变其组成以作为一种免疫疗法的努力。尽管有大量的临床前证据,但将微生物群调控方法转化为人类治疗尚未形成商业化的疗法。合成生物学方法也日益受到关注,工程化细菌癌症疗法已进入临床前和临床试验阶段。
TLSs在癌症中的预后和预测价值增强了人们将这些结构作为抗肿瘤免疫潜在介质的兴趣。尽管TLSs已在许多癌症类型中被发现,但用于定义和表征TLSs的标志物在不同研究中常常存在差异,这使比较其预测价值以及评估不同癌症类型间TLSs异质性的努力变得复杂。因此,开发测量TLSs数量和组成的标准化方法,可能会进一步揭示它们在不同疾病环境中的预测和预后价值。与此相关的是,对TLSs更全面的表征可能基于细胞组成、位置、成熟度和功能等方面,从而识别出一系列TLSs状态。类似于癌症中T细胞状态的定义极大地增进了我们对特定T细胞群在肿瘤特异性免疫中作用的理解,对TLSs状态的分子定义可能有助于提升其作为预后和预测标志物的价值。最后,为了更好地发挥其作为治疗靶点的价值,更深入地理解TLSs功能以及TLSs对自身免疫毒性的潜在贡献将非常重要。
更好地理解微生物在癌症中的作用为改善癌症护理周期的每个阶段提供了机会,但重大挑战依然存在。协调一致地努力,在采用金标准污染对照的情况下,对肿瘤、粪便和血液样本中的癌症相关微生物群进行表征,将极大地推动这一进展。这类似于癌症基因组图谱在表征癌症体细胞突变景观中的作用。目前正在进行的大规模临床试验正在测试微生物群调控方法的疗效,范围从饮食调整到瘤内注射工程化细菌。这些细菌癌症疗法如果安全有效,将极大地扩展癌症治疗的手段。总之,整合以宿主为中心和以微生物为中心的癌症视角,可能会改善患者预后,同时为癌症-宿主-微生物的演化提供细致入微的理解。
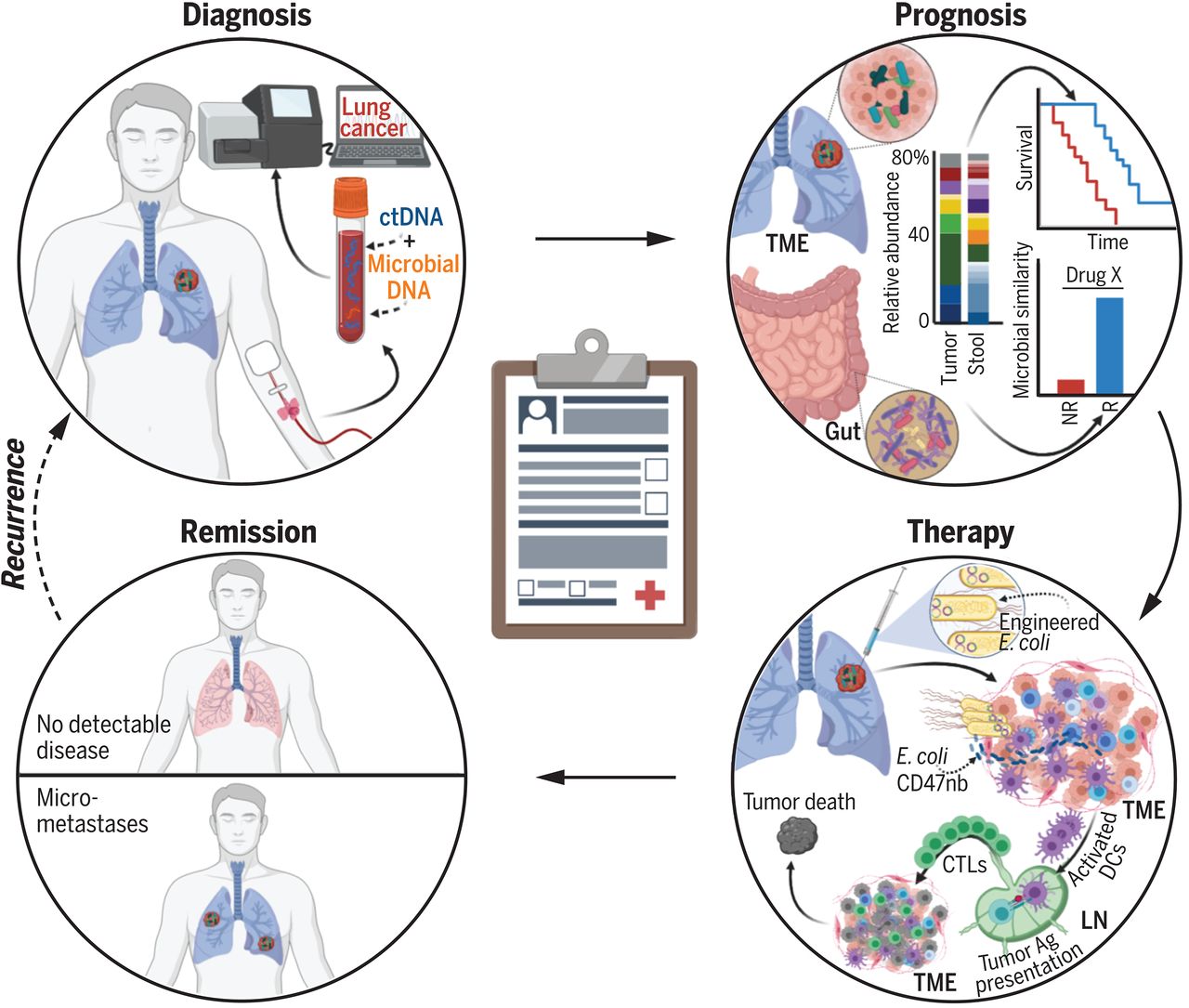
图:微生物影响癌症治疗的机会。诊断:癌症特异性血液微生物DNA可补充细胞游离肿瘤DNA(ctDNA)。 预后:肠道及肿瘤内微生物群可分层预测患者结局(NR,无应答者;R,应答者;TME,肿瘤微环境)。治疗:瘤内注射能产生CD47纳米抗体(CD47nb)的大肠杆菌,可能通过增强树突状细胞(DC)吞噬作用、淋巴结(LN)抗原(Ag)呈递及细胞毒性T淋巴细胞(CTL)活性,从而引发全身性抗肿瘤免疫。