Cyclin-dependent kinases (CDKs), in complex with their cyclin partners, modulate the transition through phases of the cell division cycle. Cyclin D–CDK complexes are important in cancer progression, especially for certain types of breast cancer. Fassl et al. discuss advances in understanding the biology of cyclin D–CDK complexes that have led to new concepts about how drugs that target these complexes induce cancer cell cytostasis and suggest possible combinations to widen the types of cancer that can be treated. They also discuss progress in overcoming resistance to cyclin D–CDK inhibitors and their possible application to diseases beyond cancer. —GKA
Cyclins and cyclin-dependent kinases (CDKs) drive cell division. Of particular importance to the cancer field are D-cyclins, which activate CDK4 and CDK6. In normal cells, the activity of cyclin D–CDK4/6 is controlled by the extracellular pro-proliferative or inhibitory signals. By contrast, in many cancers, cyclin D–CDK4/6 kinases are hyperactivated and become independent of mitogenic stimulation, thereby driving uncontrolled tumor cell proliferation. Mouse genetic experiments established that cyclin D–CDK4/6 kinases are essential for growth of many tumor types, and they represent potential therapeutic targets. Genetic and cell culture studies documented the dependence of breast cancer cells on CDK4/6. Chemical CDK4/6 inhibitors were synthesized and tested in preclinical studies. Introduction of these compounds to the clinic represented a breakthrough in breast cancer treatment and will likely have a major impact on the treatment of many other tumor types.
Small-molecule CDK4/6 inhibitors (palbociclib, ribociclib, abemaciclib) showed impressive results in clinical trials for patients with hormone receptor–positive breast cancers. Addition of CDK4/6 inhibitors to standard endocrine therapy substantially extended median progression-free survival and prolonged median overall survival. Consequently, all three CDK4/6 inhibitors have been approved for treatment of women with advanced or metastatic hormone receptor–positive breast cancers. In the past few years, the renewed interest in CDK4/6 biology has yielded several surprising discoveries. The emerging concept is that CDK4/6 kinases regulate a much wider set of cellular functions than anticipated. Consequently, CDK4/6 inhibitors, beyond inhibiting tumor cell proliferation, affect tumor cells and the tumor environment through mechanisms that are only beginning to be elucidated. For example, inhibition of CDK4/6 affects antitumor immunity acting both on tumor cells and on the host immune system. CDK4/6 inhibitors were shown to enhance the efficacy of immune checkpoint blockade in preclinical mouse cancer models. These new concepts are now being tested in clinical trials.
Palbociclib, ribociclib, and abemaciclib are being tested in more than 300 clinical trials for more than 50 tumor types. These trials evaluate CDK4/6 inhibitors in combination with a wide range of therapeutic compounds that target other cancer-relevant pathways. Several other combination treatments were shown to be efficacious in preclinical studies and will enter clinical trials soon. Another CDK4/6 inhibitor, trilaciclib, is being tested for its ability to shield normal cells of the host from cytotoxic effects of chemotherapy. New CDK4/6 inhibitors have been developed and are being assessed in preclinical and clinical trials. The major impediment in the therapeutic use of CDK4/6 inhibitors is that patients who initially respond to treatment often develop resistance and eventually succumb to the disease. Moreover, a substantial fraction of tumors show preexisting, intrinsic resistance to CDK4/6 inhibitors. One of the main challenges will be to elucidate the full range of resistance mechanisms. Even with the current, limited knowledge, one can envisage the principles of new, improved approaches to overcome known resistance mechanisms. Another largely unexplored area for future study is the possible involvement of CDK4/6 in other pathologic states beyond cancer. This will be the subject of intense studies, and it may extend the utility of CDK4/6 inhibitors to the treatment of other diseases.
Cyclin-dependent kinases 4 and 6 (CDK4 and CDK6) and their activating partners, D-type cyclins, link the extracellular environment with the core cell cycle machinery. Constitutive activation of cyclin D–CDK4/6 represents the driving force of tumorigenesis in several cancer types. Small-molecule inhibitors of CDK4/6 have been used with great success in the treatment of hormone receptor–positive breast cancers and are in clinical trials for many other tumor types. Unexpectedly, recent work indicates that inhibition of CDK4/6 affects a wide range of cellular functions such as tumor cell metabolism and antitumor immunity. We discuss how recent advances in understanding CDK4/6 biology are opening new avenues for the future use of cyclin D–CDK4/6 inhibitors in cancer treatment.
细胞周期蛋白依赖性激酶(CDKs)与其周期蛋白伴侣形成复合物,调控细胞分裂周期各阶段的转换。周期蛋白D-CDK复合物在癌症进展中具有重要作用,尤其对于某些类型的乳腺癌。Fassl等人讨论了在理解周期蛋白D-CDK复合物生物学方面的进展,这些进展带来了关于靶向这些复合物的药物如何诱导癌细胞停滞的新概念,并提出了可能扩大可治疗癌症类型的联合用药方案。他们还讨论了在克服对周期蛋白D-CDK抑制剂耐药性方面的进展,以及其在癌症以外疾病中可能的应用前景。—GKA
周期蛋白和细胞周期蛋白依赖性激酶(CDKs)驱动细胞分裂。对癌症领域尤为重要是D-周期蛋白,它们激活CDK4和CDK6。在正常细胞中,周期蛋白D–CDK4/6的活性受细胞外促增殖或抑制信号的控制。相比之下,在许多癌症中,周期蛋白D–CDK4/6激酶被过度激活,并且不依赖于有丝分裂原刺激,从而驱动肿瘤细胞不受控制地增殖。小鼠遗传实验证实,周期蛋白D–CDK4/6激酶对于许多肿瘤类型的生长至关重要,它们是潜在的治疗靶点。遗传学和细胞培养研究证实了乳腺癌细胞对CDK4/6的依赖性。化学CDK4/6抑制剂被合成并在临床前研究中进行了测试。这些化合物进入临床代表了乳腺癌治疗的突破,并可能对许多其他肿瘤类型的治疗产生重大影响。
小分子CDK4/6抑制剂(哌柏西利、瑞博西利、阿贝西利)在激素受体阳性乳腺癌患者的临床试验中显示出令人瞩目的结果。在标准内分泌治疗中加入CDK4/6抑制剂显著延长了中位无进展生存期和总生存期。因此,所有三种CDK4/6抑制剂均已被批准用于治疗晚期或转移性激素受体阳性乳腺癌女性患者。过去几年,对CDK4/6生物学重新燃起的兴趣带来了几个令人惊讶的发现。新兴的观点是,CDK4/6激酶调控的细胞功能范围远比预期的更广。因此,CDK4/6抑制剂除了抑制肿瘤细胞增殖外,还通过刚刚开始阐明的机制影响肿瘤细胞和肿瘤微环境。例如,抑制CDK4/6会影响抗肿瘤免疫,同时作用于肿瘤细胞和宿主免疫系统。临床前小鼠癌症模型显示,CDK4/6抑制剂能增强免疫检查点阻断疗法的疗效。这些新概念目前正在临床试验中进行验证。
哌柏西利、瑞博西利和阿贝西利正在超过300项针对50多种肿瘤类型的临床试验中进行测试。这些试验评估了CDK4/6抑制剂与靶向其他癌症相关通路的多种治疗化合物的联合应用。临床前研究显示其他几种联合治疗方案有效,并将很快进入临床试验。另一种CDK4/6抑制剂曲拉西利正在测试其保护宿主正常细胞免受化疗细胞毒性影响的能力。新的CDK4/6抑制剂已被开发出来,并正在接受临床前和临床试验评估。CDK4/6抑制剂治疗应用的主要障碍是,最初对治疗有反应的患者常常会产生耐药性并最终死于该疾病。此外,相当一部分肿瘤对CDK4/6抑制剂表现出预先存在的内在耐药性。主要挑战之一将是阐明所有耐药机制。即使以目前有限的知识,也可以设想出新的、改进的、克服已知耐药机制方法的原理。另一个未来研究中很大程度上尚未探索的领域是CDK4/6可能参与癌症以外的其他病理状态。这将成为深入研究的热点,并可能将CDK4/6抑制剂的用途扩展到其他疾病的治疗中。
细胞周期蛋白依赖性激酶4和6(CDK4和CDK6)及其激活伴侣D型周期蛋白,将细胞外环境与核心细胞周期机制联系起来。周期蛋白D–CDK4/6的组成性激活是几种癌症类型中肿瘤发生的驱动力。CDK4/6小分子抑制剂已成功用于治疗激素受体阳性乳腺癌,并正在许多其他肿瘤类型的临床试验中进行测试。出乎意料的是,最近的研究表明,抑制CDK4/6会影响广泛的细胞功能,例如肿瘤细胞代谢和抗肿瘤免疫。我们将讨论最近在理解CDK4/6生物学方面的进展如何为未来在癌症治疗中使用周期蛋白D–CDK4/6抑制剂开辟新途径。
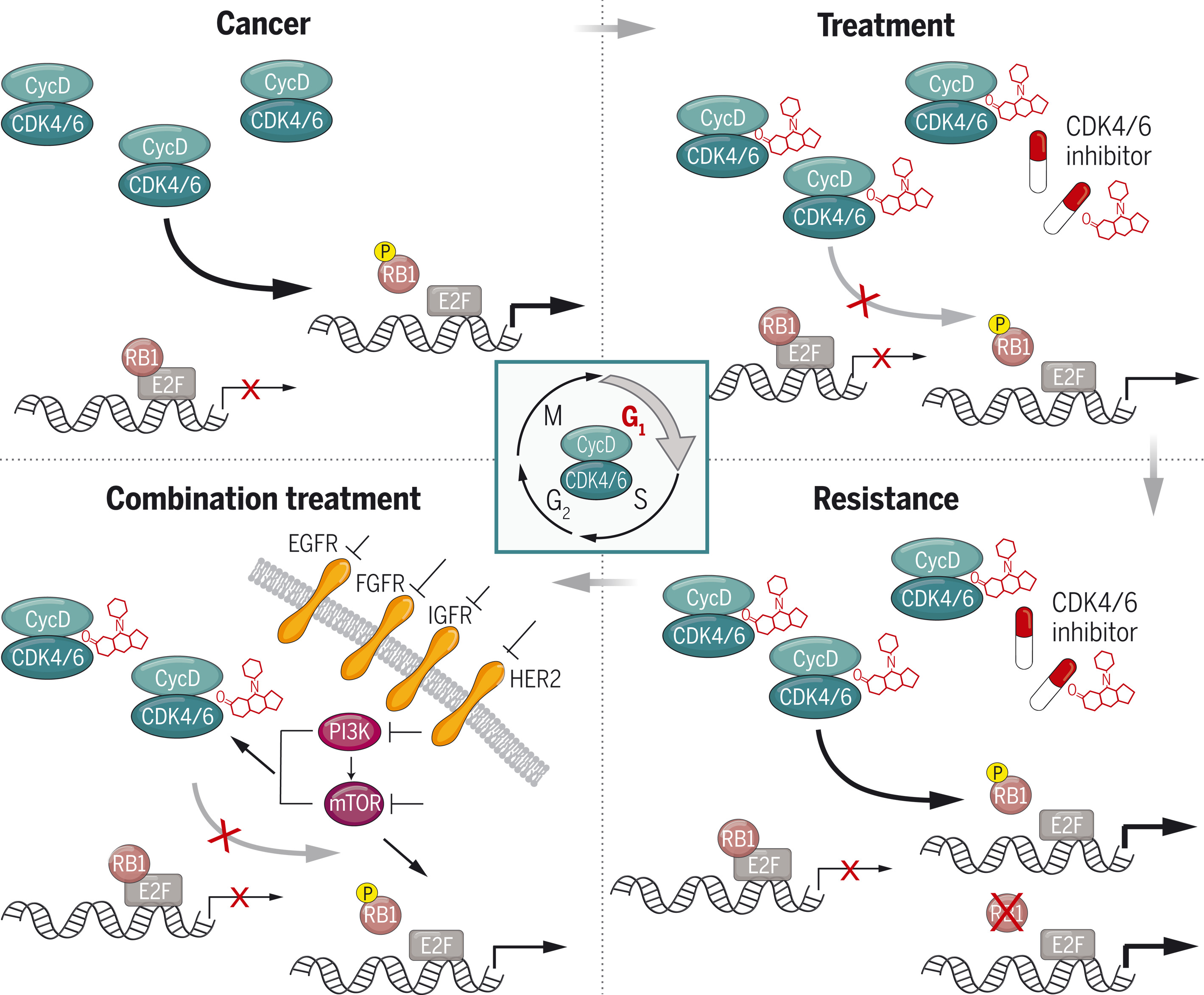
图:靶向细胞周期蛋白D–CDK4/6用于癌症治疗。D型细胞周期蛋白(CycD)在细胞周期的G1期激活CDK4和CDK6,并通过磷酸化视网膜母细胞瘤蛋白RB1促进细胞周期进程。RB1抑制E2F转录因子;RB1的磷酸化会激活E2F驱动的转录。在许多癌症中,CycD-CDK4/6持续激活并驱动不受控制的细胞增殖。小分子CDK4/6抑制剂的开发提供了一种治疗工具,可抑制持续的CycD-CDK4/6活性并阻碍癌细胞增殖。与多种靶向治疗类似,尽管使用CDK4/6抑制剂,肿瘤最终仍会产生耐药性并恢复细胞增殖。目前,涉及CDK4/6抑制剂联合其他通路抑制的新型联合疗法正在临床试验中,旨在延缓或克服耐药性。