Cancers diagnosed early are often more responsive to treatment. Blood tests that detect molecular markers of cancer have successfully identified individuals already known to have the disease. Lennon et al. conducted an exploratory study that more closely reflects the way in which such blood tests would be used in the future. They evaluated the feasibility and safety of incorporating a multicancer blood test into the routine clinical care of 10,000 women with no history of cancer. Over a 12-month period, the blood test detected 26 cancers of different types. A combination of the blood test and positron emission tomography–computed tomography (PET-CT) imaging led to surgical removal of nine of these cancers. Use of the blood test did not result in a large number of futile follow-up procedures.
The goal of earlier cancer detection is to identify the disease at a stage when it can be effectively treated, thereby offering the patient a better chance of long-term survival. Adherence to screening modalities known to decrease cancer mortality such as colonoscopy, mammography, low-dose computed tomography, and Pap smears varies widely. Moreover, the majority of cancer types are diagnosed only when symptoms occur. Multicancer blood tests offer the exciting possibility of detecting many cancer types at a relatively early stage and in a minimally invasive manner.
Evaluation of the feasibility and safety of multicancer blood testing requires prospective interventional studies. We designed such a study to answer four critical questions: (i) Can a multicancer blood test detect cancers not previously detected by other means? (ii) Can a positive test result lead to surgical intervention with curative intent? (iii) Can testing be incorporated into routine clinical care and not discourage patients from undergoing recommended screening tests such as mammography? (iv) Can testing be performed safely, without incurring a large number of unnecessary, invasive follow-up tests?
We evaluated a blood test that detects DNA mutations and protein biomarkers of cancer in a prospective, interventional study of 10,006 women who were 65 to 75 years old and who had no prior history of cancer. Positive blood tests were followed by diagnostic positron emission tomography–computed tomography (PET-CT), which served to independently confirm and precisely localize the site and extent of disease if present. The study design incorporated several features to maximize the safety of testing to the participants.
Of the 10,006 enrollees, 9911 (99.1%) could be assessed with respect to the four questions posed above. (i) Detection: Of 96 cancers incident during the study period, 26 were first detected by blood testing and 24 additional cancers by conventional screening. Fifteen of the 26 patients in whom cancer was first detected by blood testing underwent PET-CT imaging, and 11 patients developed signs or symptoms of cancer after the blood test that led to imaging procedures other than PET-CT. The specificity and positive predictive value (PPV) of blood testing alone were 98.9% and 19.4%, respectively, and combined with PET-CT, the specificity and PPV increased to 99.6% and 28.3%. The blood test first detected 14 of 45 cancers (31%) in seven organs for which no standard-of-care screening test is available. (ii) Intervention: Of the 26 cancers first detected by blood testing, 17 (65%) had localized or regional disease. Of the 15 participants with positive blood tests as well as positive PET-CT scans, 9 (60%) underwent surgery with curative intent. (iii) Incorporation into clinical care: Blood testing could be combined with conventional screening, leading to detection of more than half of the total incident cancers observed during the study period. Blood testing did not deter participants from undergoing mammography, and surveys revealed that 99% of participants would join a similar, subsequent study if offered. (iv) Safety: 99% of participants did not require any follow-up of blood testing results, and only 0.22% underwent an unnecessary invasive diagnostic procedure as a result of a false-positive blood test.
A minimally invasive blood test in combination with PET-CT can safely detect and precisely localize several types of cancers in individuals not previously known to have cancer, in some cases enabling treatment with intent to cure. Further studies will be required to assess the clinical utility, risk-benefit ratio, and cost-effectiveness of such testing.
Cancer treatments are often more successful when the disease is detected early. We evaluated the feasibility and safety of multicancer blood testing coupled with positron emission tomography–computed tomography (PET-CT) imaging to detect cancer in a prospective, interventional study of 10,006 women not previously known to have cancer. Positive blood tests were independently confirmed by a diagnostic PET-CT, which also localized the cancer. Twenty-six cancers were detected by blood testing. Of these, 15 underwent PET-CT imaging and nine (60%) were surgically excised. Twenty-four additional cancers were detected by standard-of-care screening and 46 by neither approach. One percent of participants underwent PET-CT imaging based on false-positive blood tests, and 0.22% underwent a futile invasive diagnostic procedure. These data demonstrate that multicancer blood testing combined with PET-CT can be safely incorporated into routine clinical care, in some cases leading to surgery with intent to cure.
早期诊断出的癌症通常对治疗反应更好。能够检测癌症分子标志物的血液检测已成功识别出已知患有该疾病的个体。Lennon等人进行了一项探索性研究,更贴近未来此类血液检测的实际应用方式。他们评估了将多癌种血液检测纳入10,000名无癌症史女性的常规临床护理中的可行性和安全性。在12个月的时间内,该血液检测检测出了26种不同类型的癌症。血液检测与正电子发射断层扫描-计算机断层扫描(PET-CT)成像相结合,使得其中9种癌症得以手术切除。使用血液检测并未导致大量无效的后续检查程序。
早期癌症检测的目标是在疾病可以有效治疗的阶段发现它,从而为患者提供更好的长期生存机会。对于已知可降低癌症死亡率的筛查方式,如结肠镜检查、乳腺X线摄影、低剂量计算机断层扫描和巴氏涂片,人们的依从性差异很大。此外,大多数癌症类型仅在出现症状时才被诊断出来。多癌种血液检测提供了以相对早期和微创方式检测多种癌症类型的激动人心的可能性。
评估多癌种血液检测的可行性和安全性需要前瞻性干预研究。我们设计了这样一项研究来回答四个关键问题:(i)多癌种血液检测能否检测出先前未通过其他方式检测到的癌症?(ii)阳性检测结果能否导致旨在治愈的手术干预?(iii)检测能否纳入常规临床护理,并且不阻止患者接受推荐的筛查测试(如乳腺X线摄影)?(iv)检测能否安全进行,而不会引发大量不必要的侵入性后续检查?
我们在一项前瞻性干预研究中评估了一种检测癌症DNA突变和蛋白质生物标志物的血液检测,研究对象为10,006名年龄在65至75岁之间且无癌症史的女性。阳性血液检测后进行了诊断性正电子发射断层扫描-计算机断层扫描(PET-CT),用于独立确认并精确定位疾病(如果存在)的部位和范围。研究设计包含多个特征,以最大限度地提高参与者检测的安全性。
在10,006名入组者中,有9,911名(99.1%)可以针对上述四个问题进行评估。(i)检测:在研究期间发生的96例癌症中,有26例首次通过血液检测发现,另有24例通过常规筛查发现。在首次通过血液检测发现癌症的26名患者中,有15名接受了PET-CT成像,11名患者在血液检测后出现癌症体征或症状,从而进行了PET-CT以外的影像学检查。单独血液检测的特异性和阳性预测值(PPV)分别为98.9%和19.4%,与PET-CT联合后,特异性和PPV提高到99.6%和28.3%。在7个没有标准筛查检测的器官中,血液检测首次检测出45例癌症中的14例(31%)。(ii)干预:在首次通过血液检测发现的26例癌症中,17例(65%)为局限性或区域性疾病。在血液检测阳性且PET-CT扫描阳性的15名参与者中,9名(60%)接受了旨在治愈的手术。(iii)纳入临床护理:血液检测可与常规筛查相结合,检测出研究期间观察到的总癌症病例的一半以上。血液检测并未阻止参与者接受乳腺X线摄影,调查显示,99%的参与者如果被邀请,会参加类似的后续研究。(iv)安全性:99%的参与者不需要对血液检测结果进行任何随访,只有0.22%因假阳性血液检测结果而接受了不必要的侵入性诊断程序。
微创血液检测与PET-CT相结合,可以安全地检测并精确定位先前未知患有癌症的个体中的几种癌症类型,在某些情况下能够实现旨在治愈的治疗。需要进一步的研究来评估此类检测的临床实用性、风险-收益比和成本效益。
当疾病被早期发现时,癌症治疗往往更成功。我们在一项前瞻性干预研究中评估了多癌种血液检测联合正电子发射断层扫描-计算机断层扫描(PET-CT)成像检测癌症的可行性和安全性,研究对象为10,006名先前未知患有癌症的女性。阳性血液检测结果由诊断性PET-CT独立确认,该检查还能定位癌症。血液检测检测出26例癌症。其中15例接受了PET-CT成像,9例(60%)接受了手术切除。另外24例癌症通过标准筛查检测发现,46例通过两种方法均未发现。1%的参与者因假阳性血液检测结果接受了PET-CT成像,0.22%接受了无效的侵入性诊断程序。这些数据表明,多癌种血液检测与PET-CT联合可以安全地纳入常规临床护理,在某些情况下可导致旨在治愈的手术。
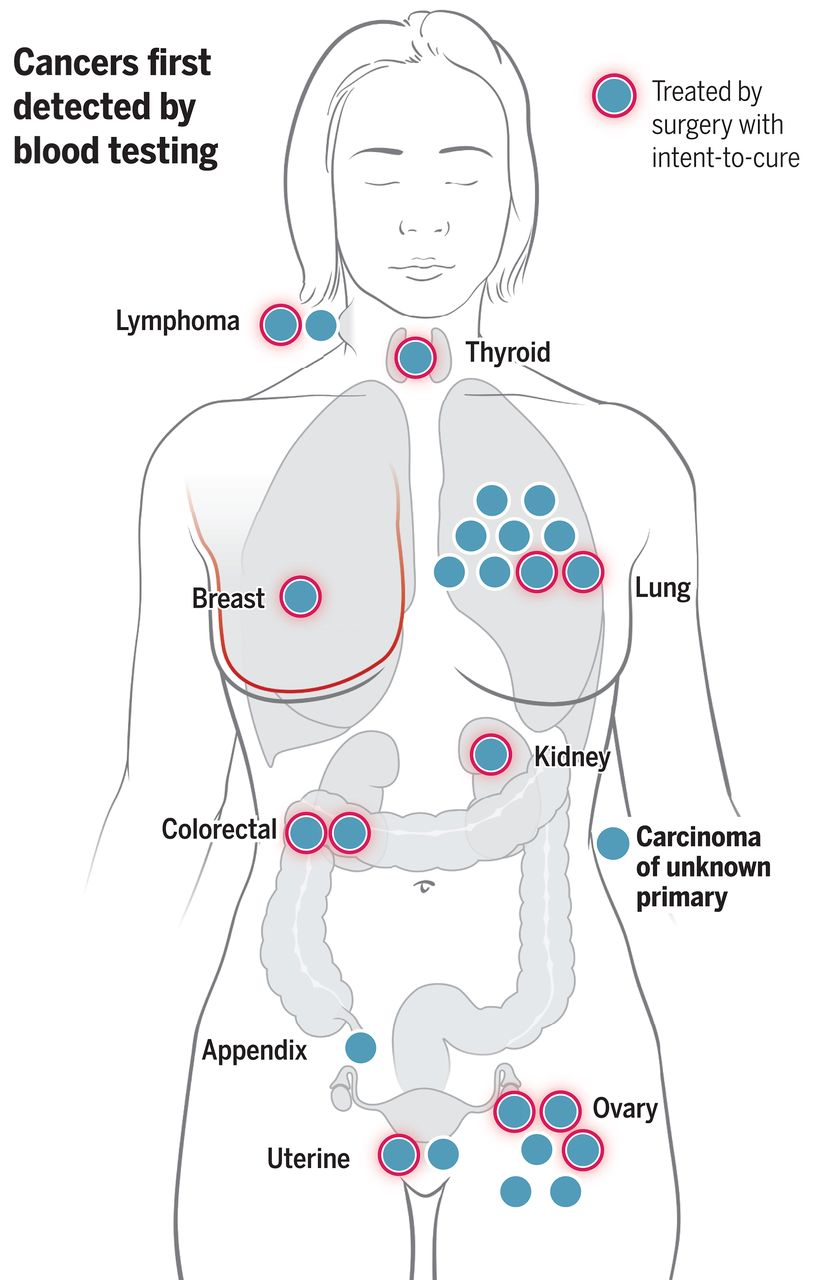
图:通过血液检测发现的癌症概述。血液检测首次发现了10个器官中的26例癌症(蓝点)。带有红色光晕的蓝点代表26例癌症中接受治愈性意图手术治疗的12例。这12例中有9例是通过血液检测联合PET-CT发现的,其余3例通过血液检测联合其他影像学检查方式确认。
Feasibility of blood testing combined with PET-CT to screen for cancer and guide intervention