Circadian clocks help to coordinate physiological processes with the daily cycles of light and dark and periods of feeding, activity, and rest. Being out of sync with such 24-hour cycles can have unhealthy effects. Sancar and Van Gelder review the available evidence regarding circadian disruption and predisposition to cancer and circadian variations in response to cancer chemotherapy. The literature can be difficult to interpret. For example, complete knockouts of clock genes are not the same as shift work. Overall, they find that the jury is still out on whether circadian disruption can promote cancer in general and if the timing of cancer treatment can be optimized. However, enough indications are present that further research is recommended.
The core of the mammalian circadian clock mechanism is a time-delayed transcription-translation feedback loop (TTFL), which influences the transcription and expression of a large fraction of the transcriptome. Through this mechanism, the mammalian circadian clock modulates many physiological functions, including the timing of cell division and rates of metabolism in specific tissues. Circadian clock dysfunction is associated with several human disease states, including jet lag and sleep phase disorders, and it likely contributes significantly to the development of metabolic syndrome. With respect to cancer, animal studies have suggested that specific carcinogenic mechanisms, such as ultraviolet radiation for skin cancer, have a strong circadian rhythm. Epidemiologic studies have yielded conflicting results as to whether circadian clock disruption by night or shift work is carcinogenic. In animal studies, tumors grafted into animals with disrupted rhythms grow more rapidly than those grafted into control animals. Studies of mice genetically lacking specific components of the circadian clock show increased rates of tumorigenesis for certain clock genes and certain tumors but show reduced rates for other clock genes. Similarly, the response to chemotherapy may also vary with time of day, which has led to enthusiasm for chronochemotherapy as a means to improve the therapeutic efficacy of cancer treatment while limiting toxicity. However, clinical trials of chronochemotherapy have generally not shown improved efficacy and have even shown worse outcomes in subsets of patients compared with conventionally timed therapies.
Polymorphisms in circadian clock genes including Npas2 and Clock have been identified in genome-wide association studies as relatively weak but significant modifiers of breast cancer incidence, and core circadian clock gene expression is frequently dysregulated in human tumors. However, it is not possible to generalize that loss of the clock leads to increased cancer incidence, as some clockless animals actually show resistance to specific cancer pathways (e.g., Cryptochrome-less mice are resistant to p53 mutation–induced tumors). In other cases, different clock gene mutations result in opposite phenotypes with respect to carcinogenesis for the same tumor type. Perhaps the best-studied mechanistic interaction between circadian clock and carcinogenesis involves studies of the circadian rhythms of nucleotide excision DNA repair. Although basal excision repair has a circadian rhythm with a specific maximal phase, the rhythm of an individual gene’s repair is dependent on the phase of that gene’s transcriptional rhythm; there is no single phase at which DNA is generally more or less easily repaired. Other notable advances in the field include the demonstration of direct mechanistic linkage of c-MYC expression to circadian clock control and the demonstration that oncogenes c-Myc, p53, and Ras all affect the circadian core TTFL, consistent with the finding that the circadian clock of tumors is frequently dysregulated.
Tumorigenesis is clearly affected by circadian mechanisms, but the hypothesis that circadian clock genes are general tumor suppressors is not supported. Rather, specific tumors and their underlying mechanisms are differentially affected by the function of specific clock genes. Conversely, specific oncogenes may cause dysregulation of the circadian clock in tumors; the pathogenic significance of the dysregulated clock in tumors is not fully understood. The example of circadian control of DNA nucleotide excision repair illuminates the challenges in exploiting the interaction between clocks and cancer clinically, as the phase of circadian susceptibility to DNA damage varies for each gene on the basis of its underlying transcriptional rhythm. Although the concept of chronochemotherapy is attractive, the complexities of clock-cancer interactions make prediction of the effects of timed drug administration challenging. Mistiming of chemotherapeutic agents has the potential to be harmful. As chemotherapeutic agents increase in specificity, the circadian effects of administration may be better understood and optimized by understanding the specific interactions between the circadian clock mechanism and therapeutic targets.
The circadian clock coordinates daily rhythmicity of biochemical, physiologic, and behavioral functions in humans. Gene expression, cell division, and DNA repair are modulated by the clock, which gives rise to the hypothesis that clock dysfunction may predispose individuals to cancer. Although the results of many epidemiologic and animal studies are consistent with there being a role for the clock in the genesis and progression of tumors, available data are insufficient to conclude that clock disruption is generally carcinogenic. Similarly, studies have suggested a circadian time-dependent efficacy of chemotherapy, but clinical trials of chronochemotherapy have not demonstrated improved outcomes compared with conventional regimens. Future hypothesis-driven and discovery-oriented research should focus on specific interactions between clock components and carcinogenic mechanisms to realize the full clinical potential of the relationship between clocks and cancer.
昼夜节律钟有助于协调生理过程与每日的光暗周期以及进食、活动和休息时段。与这种24小时周期不同步可能会产生不健康的影响。Sancar和Van Gelder回顾了关于昼夜节律紊乱与癌症易感性以及癌症化疗反应中昼夜节律变化的现有证据。相关文献可能难以解释。例如,时钟基因的完全敲除与轮班工作并不相同。总体而言,他们认为关于昼夜节律紊乱是否普遍促进癌症发生以及癌症治疗时机是否可以优化的问题,尚无定论。然而,有足够的迹象表明,建议进行进一步研究。
哺乳动物昼夜节律钟机制的核心是一个延时转录-翻译反馈回路(TTFL),它影响转录组中很大一部分的转录和表达。通过这一机制,哺乳动物昼夜节律钟调节许多生理功能,包括细胞分裂的时机和特定组织中的代谢速率。昼夜节律钟功能障碍与多种人类疾病状态相关,包括时差反应和睡眠时相障碍,并且可能显著促进代谢综合征的发展。关于癌症,动物研究表明,特定的致癌机制,如紫外线辐射导致皮肤癌,具有强烈的昼夜节律。关于夜间工作或轮班工作导致的昼夜节律钟紊乱是否致癌,流行病学研究得出了相互矛盾的结果。在动物研究中,将肿瘤移植到节律紊乱的动物体内,其生长速度比移植到对照动物体内更快。对基因上缺乏特定昼夜节律钟组件的小鼠的研究显示,对于某些时钟基因和某些肿瘤,肿瘤发生率增加,但对于其他时钟基因则显示发生率降低。同样,对化疗的反应也可能随一天中的时间而变化,这引起了人们对时间化学疗法作为一种在限制毒性的同时提高癌症治疗效果的手段的热情。然而,时间化学疗法的临床试验通常未显示出改善的疗效,甚至在某些患者亚组中显示出比常规时间疗法更差的结果。
全基因组关联研究已经确定,包括Npas2和Clock在内的昼夜节律钟基因的多态性是乳腺癌发病率的相对较弱但显著的修饰因子,并且核心昼夜节律钟基因的表达在人类肿瘤中经常失调。然而,不能一概而论地认为时钟功能的丧失会导致癌症发病率增加,因为一些无时钟动物实际上对特定的癌症通路表现出抵抗力(例如,缺乏隐花色素的小鼠对p53突变诱导的肿瘤具有抵抗力)。在其他情况下,不同的时钟基因突变导致同一肿瘤类型在致癌作用方面出现相反的表型。也许研究得最深入的昼夜节律钟与致癌作用之间的机制相互作用涉及对核苷酸切除DNA修复的昼夜节律的研究。尽管基础切除修复具有一个特定的最大时相的昼夜节律,但单个基因修复的节律取决于该基因转录节律的时相;没有一个单一的时相使得DNA普遍更容易或更难修复。该领域其他值得注意的进展包括证明了c-MYC表达与昼夜节律钟控制的直接机制联系,以及证明了癌基因c-Myc、p53和Ras都影响昼夜节律核心TTFL,这与肿瘤的昼夜节律钟经常失调的发现一致。
肿瘤发生明显受昼夜节律机制影响,但昼夜节律钟基因是普遍肿瘤抑制因子的假设并未得到支持。相反,特定肿瘤及其潜在机制受到特定时钟基因功能的差异性影响。反之,特定的癌基因可能导致肿瘤中昼夜节律钟的失调;肿瘤中失调时钟的致病意义尚不完全清楚。DNA核苷酸切除修复的昼夜节律控制的例子阐明了在临床上利用时钟与癌症之间相互作用的挑战,因为对DNA损伤的昼夜节律易感性的时相根据每个基因的潜在转录节律而变化。虽然时间化学疗法的概念很有吸引力,但时钟-癌症相互作用的复杂性使得预测定时给药的效果具有挑战性。化疗药物的时间不当可能有害。随着化疗药物特异性的提高,通过了解昼夜节律钟机制与治疗靶点之间的特定相互作用,可以更好地理解和优化给药的昼夜节律效应。
昼夜节律钟协调人类生化、生理和行为功能的每日节律性。基因表达、细胞分裂和DNA修复受时钟调节,这引发了时钟功能障碍可能使个体易患癌症的假说。尽管许多流行病学和动物研究的结果与时钟在肿瘤发生和进展中发挥作用是一致的,但现有数据不足以得出时钟紊乱普遍致癌的结论。同样,研究表明化疗具有昼夜节律时间依赖性疗效,但时间化学疗法的临床试验并未显示出比常规方案改善的结果。未来的假设驱动和发现导向研究应侧重于时钟组件与致癌机制之间的特定相互作用,以实现时钟与癌症关系的全部临床潜力。
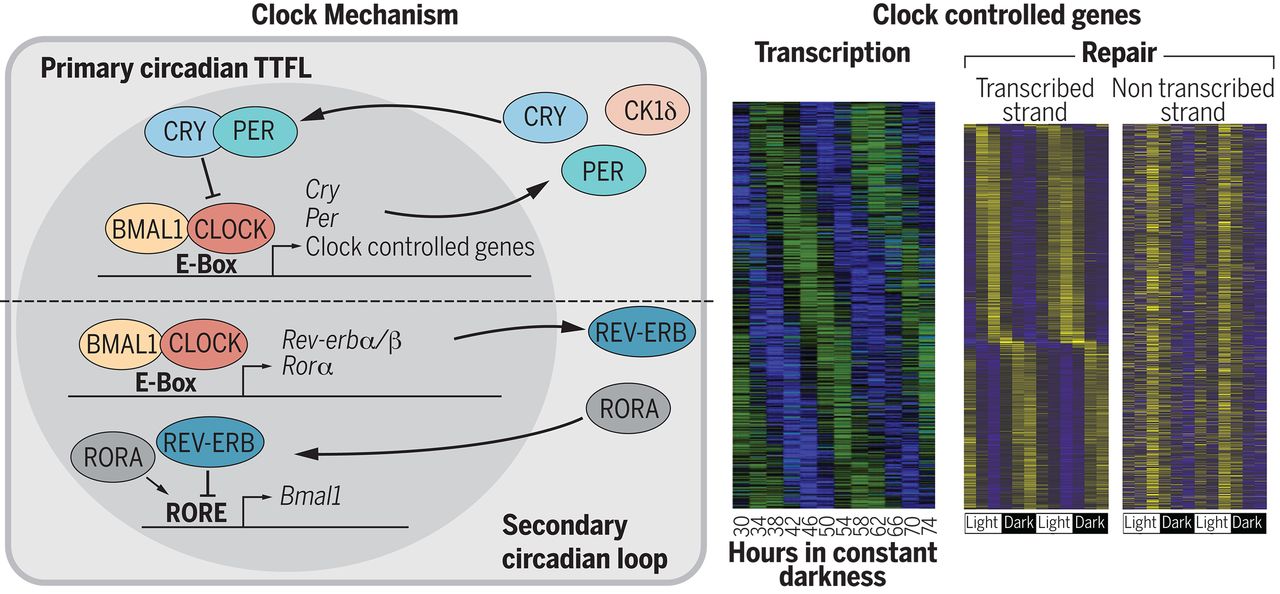
图:哺乳动物生物钟调控转录过程及DNA修复机制。(左侧)哺乳动物生物钟机制是一个具备时间延迟效应的转录-翻译反馈回路(TTFL)。BMAL1-CLOCK构成正调控臂,而隐花色素(CRY1和CRY2)与周期蛋白(PER1和PER2)构成抑制性负调控臂;主反馈环路通过由REV-ERBα抑制剂和RORα激活剂组成的次级环路得以强化。在特定组织中,约10%的基因表现出显著的昼夜节律(近24小时)周期性表达。(右侧)两个热图展示了生物钟对小鼠转录过程及核苷酸切除修复的影响,其中绿色(转录)和黄色(修复)代表信号强度。左侧表达热图显示在持续黑暗44小时的小鼠肝脏中检测到854个受生物钟调控的转录本[改编自B. H. Miller等,《美国国家科学院院刊》104, 3342 (2007),版权所有(2007)美国国家科学院]。这些基因均在特定日间时段达到表达峰值。右侧修复热图则显示在12小时光照-12小时黑暗条件下经化疗药物顺铂处理后的小鼠肾脏中检测到1661个基因[改编自Y. Yang等,《美国国家科学院院刊》115, E4777 (2018),版权所有(2018)美国国家科学院]。研究分别量化了转录链(TS)和非转录链(NTS)的损伤程度。非转录链显示所有基因呈现单相节律,损伤修复峰值出现在傍晚时分。而转录链中,每个基因在周期内对应其转录峰值相位时表现出特异的修复高峰。这种个体化基因修复时序的复杂性,为优化化疗药物给药的昼夜节律时机选择带来了重大挑战。